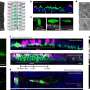
Protocol outlines steps for extracting ample DNA from uncultured microbes

Characterizing uncultured microbes starts with isolating and sequencing enough DNA from an environmental sample that may be as small as a single cell.
The protocol has been successfully employed to amplify the genomes of 201 single cells from uncultured microbes in a microbial dark matter study that was published in a July 2013 issue of Nature.
Though microbiologists say that there are more microbes in, on and around the Earth than there are stars in the galaxy, only a tiny fraction of these bacteria and archaea are known. One of the challenges scientists face in studying them is that most microbes don't thrive outside of their natural environments. Two methods used to learn more about such microbes are metagenomics and single-cell genomics. In July 2013, a Nature study led by U.S. Department of Energy Joint Genome Institute (DOE JGI) researchers employed single-cell genomics to provide information on 201 previously uncultured microbes collected from nine sampling sites around the world. The information generated shed insight on 29 "mostly uncharted" branches on the tree of life.
The technique employed to extract and amplify the DNA from single microbial cells for the microbial dark matter study is now detailed in a Nature Protocols report published ahead online April 10, 2014. The DOE JGI team describes the steps they employed from sample preservation and preparation, to single cell separation through fluorescence-activated cell sorting (FACS), to cell lysis and ultimately, whole genome amplification.
The team reported that the procedure takes four days and yields up to a microgram of DNA per single microbial cell, an amount considered "suitable" for PCR amplification and shotgun sequencing. They also noted that though the protocol was applied to microbial cells, it can also be adapted for microbial eukaryotes and mammalian cells.
More information: Rinke C et al. "Obtaining genomes from uncultivated environmental microorganisms using FACS-based single-cell genomics." Nat Protoc. 2014 May;9(5):1038-48. DOI: 10.1038/nprot.2014.067. Epub 2014 Apr 10.
Journal information: Nature Protocols , Nature
Provided by US Department of Energy