New stable isotope analysis questions the origin of black carbon in the ocean

We can probably all agree that the deep sea is not on fire.
But the charred, powdery soot that results from burning things like forests or fossil fuels makes up an important part of the carbon dissolved in the ocean. So how in the world did that soot, which scientists call black carbon, get there?
Thanks to charcoal found in the fossil record, scientists know that wildfires have happened for millions of years, since the first trees appeared. The soot that's left over—black carbon—makes up about 10% of all the carbon in Earth's soil, and until recently, scientists thought that rivers were washing it into the ocean.
Aron Stubbins, an associate professor of marine and environmental sciences at Northeastern, teamed up with a group of researchers who tested the long-established idea that rivers eventually transport black carbon into the ocean.
The team's findings, published in Nature Communications, show that the dissolved black carbon found in rivers doesn't match the readings of black carbon dwelling in the ocean.
The current study follows up on research Stubbins did in 2012, which revealed that if black carbon in the ocean is exposed to sunlight, it can turn quickly into carbon dioxide, which plays an important role in controlling Earth's climate by trapping heat. That's one of the reasons why it is important to know what happens to black carbon from the beginning, as leftover char on land, until it makes it to the ocean, Stubbins says.
"It's a lot of carbon," says Stubbins, who is also an associate professor of chemistry and chemical biology, and civil and environmental engineering at Northeastern. "We want to understand how it cycles to understand if it will ever end up in the atmosphere as carbon dioxide."
The team's work off the coasts of Savannah, Georgia, had already hinted at differences between black carbon in rivers and the ocean in 2017. Now, the idea was to test black carbon dissolved in rivers and oceans at a global scale, Stubbins says. To do that, they would need to test waters far offshore, and as deep as possible.
The team sampled the North Pacific and North Atlantic oceans, and was led by Sasha Wagner, a former postdoctoral researcher at Northeastern who is now an assistant professor of earth and environmental sciences at Rensselaer Polytechnic Institute in Troy, New York. The analysis also included the Amazon, the Congo, and other major rivers.
Near the surface and at several depths, the team probed nearly pure oceanic water, Stubbins says. If you dive 3000 meters deep (about 2 miles) into the waters of Hawaii, for example, you will be swimming in water that hasn't seen the surface of the ocean for hundreds of years. That same water hasn't been near land for even longer.
"When we went to the black carbon in all those oceans samples [at different depths], we found that they had a fairly consistent signature with one another," Stubbins says. "But they were really different from river water samples."
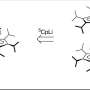
Spotting those differences was possible with analysis of stable carbon isotopes, non-radioactive forms of the same atom that have different numbers of neutrons but the same chemical properties.
Stable carbon isotopes can be used to trace things found in nature, such as black carbon, back to their origins. On land, plants mark carbon particles with a different isotopic signature than phytoplankton in the ocean. When carbon dioxide is absorbed from the air and enters terrestrial plants, they integrate carbon-12, the most abundant carbon on Earth, more efficiently than carbon-13.
"Essentially, the C-13 is a bit bigger, a bit heavier, a bit slower," Stubbins says. "So, it gets fractionated, and it is left behind."
But phytoplankton don't discriminate quite as much against the extra neutron in carbon-13, which is a much less abundant stable isotope. That's why carbon in phytoplankton and the things that eat them in the ocean have a slightly higher content of carbon-13 isotopes than trees (and the things that eat them) on land, Stubbins says. Analysis of this relative abundance, he says, can determine whether black carbon was made by trees or phytoplankton.
"By looking at the isotopes of black carbon, we found that black carbon in the oceans is not from the same source as the black carbon in rivers," he says.
It's an unexpected mystery from the deep sea: Where did the black carbon dissolved in the ocean come from?
"Because we know the ocean doesn't burn very regularly," Stubbins quips.
The next step, he says, could be focusing on matching oceanic black carbon with the isotopic signatures of other atmospheric particles, or thinking of other ways in which this carbon could end up in those waters.
"The bottom of the ocean has large stores of organic carbon that may have been processed differently in the past," he says. "Maybe somewhere in there there's a clue for the source of this material."
More information: Sasha Wagner et al. Isotopic composition of oceanic dissolved black carbon reveals non-riverine source, Nature Communications (2019). DOI: 10.1038/s41467-019-13111-7
Journal information: Nature Communications
Provided by Northeastern University