Salt helps proteins move on down the road
With a lot of hard work and a dash of salt, Rice University scientists have taken a step toward simplifying drug manufacture.
Rice chemist Christy Landes and her colleagues reported in the Proceedings of the National Academy of Sciences their strategy to make polymer membrane-based separation of proteins more efficient.
"It costs about $3 billion to bring a drug based on a biological protein to the consumer," Landes said. "And around half of that can be because purification is done by trial and error. Billions of dollars are wasted every year because there is no way to predictively design a separation scheme for a protein."
The Rice lab is developing models to predict how tuning the behavior of individual proteins at membrane interfaces will affect separation.
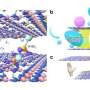
Through their research, they discovered how using salt to tune two distinct interactions between a nylon stationary phase support and a model protein, transferrin, helps make separations more efficient.
The researchers focused on salt, because "salting out" is a common step in chromatography, an industry-standard process by which the elements in a solution are separated, or "purified." The filters can be natural material like soil, absorbents like cellulose or, increasingly, polymers, including nylon.
"Think of these filters like stops along a highway," said co-author Logan Bishop, who combined his simulations with experiments by lead author Nicholas Moringo. Both are National Science Foundation (NSF) graduate fellows at Rice.
"The first stop separates out the big rigs, the next stop gets the pickup trucks, and eventually you're just left with the normal cars you want," Bishop said. "Here, we're talking about all the different forces that separate different components as a mixture moves through the column."
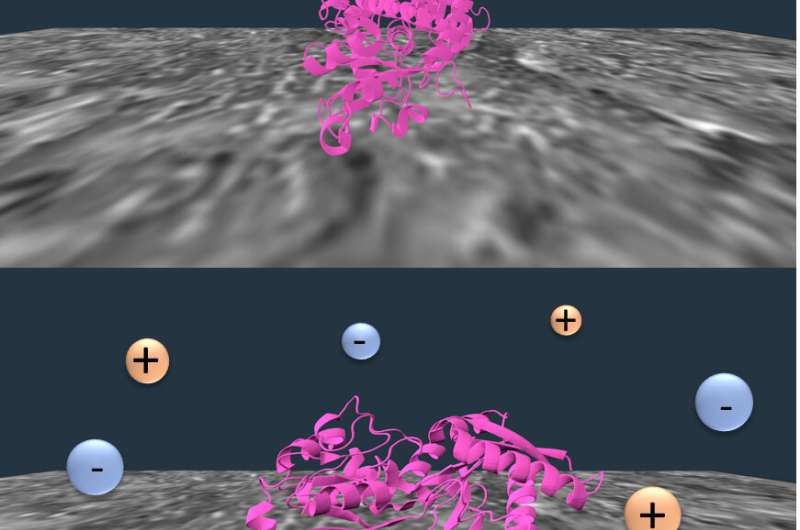
Dissolved salt creates solvated ions that interact with proteins and tune them to either stop and interact with the chromatography column or move on through the column. At the end of salting out, the desired protein can be extracted from the column with a solvent and made available for further purification steps.
Exactly how the salt influences the separation is just one question the researchers hope to answer through their experiments and simulations. "The most important thing that we did in this paper was to marry observations of individual proteins interacting at the nylon interface to an understanding of exactly how they interact," Landes said. "Our simulations now let us predict the improved separation efficiencies under realistic conditions."
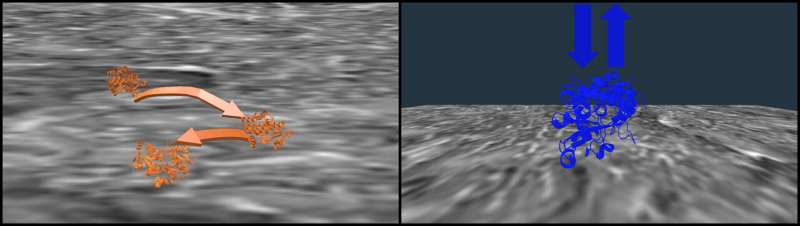
The researchers identified competing forces at the nylon surface that could be tuned by salt concentration. Observations revealed that folded transferrin proteins tended to hop around the nylon, but they partially unfold once attached to the membrane. Higher salt concentrations unfold them even more, decreasing the hopping and allowing the membrane interactions to improve the separation efficiency.

"The salt tunes the distribution of these two ways of interacting, and it also changes the structure of the protein at the interface," Landes said. "But they're each only part of the competition at the micro scale that give you the macroscopic effect. That's why it's so expensive to optimize the process with trial and error."
"We'd like to be able to test a library of surface chemistries in differing conditions in one shot on a cover slip, so we can identify the ideal conditions for separation," Moringo said. "Then we can use the simulation to predict which combinatorial answer on your chip is going to be the right one to optimize separation."
It will take years before simulations incorporate all the possible parameters, but it's a journey worth taking to make drug design and manufacture better, Bishop said. "The model is not quite complex enough, and there's an argument that we're never going to be able to match the complexity of the chromatography process," he said. "But our hope is we can get a close-enough approximation to start shaving off some of those costs and get closer to a real solution."
Landes noted that pharmaceutical companies have mastered their current techniques, as far as they go. "Nobody knows better than industrial engineers how to optimize a process to get the most results for the least amount of money, as long as they stay on the path they've been following for 70 years," she said. "But we're moving onto a transformative path. That's what academic research is for."
More information: Nicholas A. Moringo el al., "A mechanistic examination of salting out in protein–polymer membrane interactions," PNAS (2019). www.pnas.org/cgi/doi/10.1073/pnas.1909860116
Journal information: Proceedings of the National Academy of Sciences
Provided by Rice University