Science provides new way to peer into pores
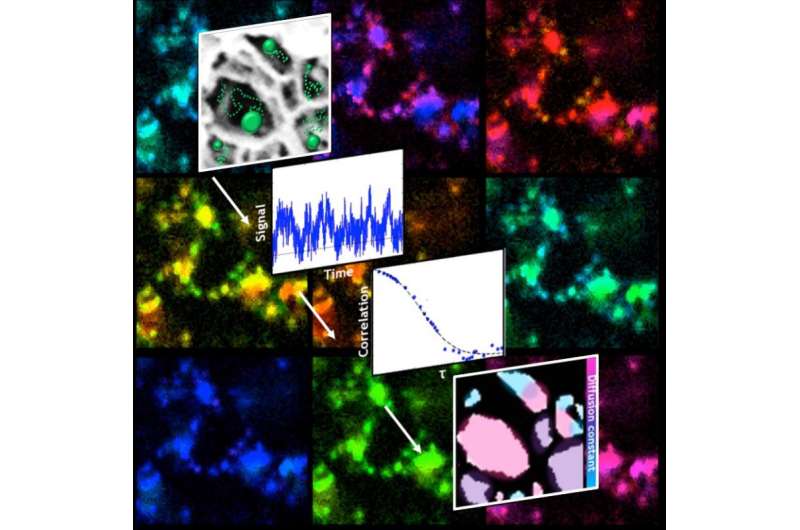
Rice University scientists led a project to "see" and measure the space in porous materials, even if that space is too small or fragile for traditional microscopes.
The Rice lab of chemist Christy Landes invented a technique to characterize such nanoscale spaces, an important advance toward her group's ongoing project to efficiently separate "proteins of interest" for drug manufacture. It should also benefit the analysis of porous materials of all kinds, like liquid crystals, hydrogels, polymers and even biological substances like cytosol, the compartmentalized fluids in cells.
The research with collaborators at the University of California, Los Angeles (UCLA) and Kansas State University appears in the American Chemical Society journal ACS Nano.
It's easy to use a fluorescent chemical compound to tag, or "label," a material and take a picture of it, Landes said. "But what if the thing you want a picture of is mostly nothing? That's the problem we had to solve to understand what was going on in the separation material."
The team aims to improve protein separation in a process called chromatography, in which solutions flow through porous material in a column. Because different materials travel at different speeds, the components separate and can be purified.
"We learned that in agarose, a porous material used to separate proteins, the clustering of charges is very important," Landes said. While the protein project succeeded, "when we matched experimental data to our theory, there was something additional contributing to the separation that we couldn't explain."
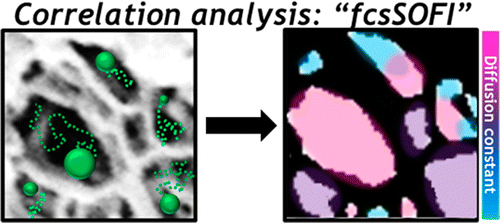
The answer appeared to be with how charged particles like nanoscale ligands arranged themselves in the pores. "It was the only possible explanation," Landes said. "So we needed a way to image the pores."
Standard techniques like atomic force, X-ray and electron microscopy would require samples to be either frozen or dried. "That would either shrink or swell or change their structures," she said.
It occurred to the team to combine their experience with the Nobel Prize-winning super-resolution microscopy and fluorescence correlation spectroscopy techniques. Super-resolution microscopy is a way to see objects at resolutions below the diffraction limit, which restricts the viewing of things that are smaller than the wavelength of light directed at them.
Correlation spectroscopy is a way to measure fluorescent particles as they fluctuate. By crunching data collected via a combination of super-resolution microscopy and correlation spectroscopy, the researchers mapped slices of the material to see where charged particles tended to cluster.
The combined technique, which they call fcsSOFI (for "fluorescence correlation spectroscopy super-resolution optical fluctuation imaging"), measures fluorescent tags as they diffuse in the pores, which allows researchers to simultaneously characterize dimensions and dynamics within the pores. The lab tested its technique on both soft agarose hydrogels and lyotropic liquid crystals. Next, they plan to extend their mapping to three-dimensional spaces.
"We now have both pieces of our puzzle: We can see our proteins interacting with charges within our porous material, and we can measure the pores," Landes said. "This has direct relevance to the protein separation problem for the $100 billion pharmaceutical industry."
More information: Characterization of Porous Materials by Fluorescence Correlation Spectroscopy Super-resolution Optical Fluctuation Imaging, ACS Nano, Article ASAP. DOI: 10.1021/acsnano.5b03430
Journal information: ACS Nano
Provided by Rice University