New process could make hydrogen peroxide available in remote places
Hydrogen peroxide, a useful all-purpose disinfectant, is found in most medicine cabinets in the developed world. But in remote villages in developing countries, where it could play an important role in health and sanitation, it can be hard to come by.
Now, a process developed at MIT could lead to a simple, inexpensive, portable device that could produce hydrogen peroxide continuously from just air, water, and electricity, providing a way to sterilize wounds, food-preparation surfaces, and even water supplies.
The new method is described this week in the journal Joule in a paper by MIT students Alexander Murray, Sahag Voskian, and Marcel Schreier and MIT professors T. Alan Hatton and Yogesh Surendranath.
Even at low concentrations, hydrogen peroxide is an effective antibacterial agent, and after carrying out its sterilizing function it breaks down into plain water, in contrast to other agents such as chlorine that can leave unwanted byproducts from its production and use.
Hydrogen peroxide is just water with an extra oxygen atom tacked on—it's H2O2, instead of H2O. That extra oxygen is relatively loosely bound, making it a highly reactive chemical eager to oxidize any other molecules around it. It's so reactive that in high concentrations it can be used as rocket fuel, and even concentrations of 35 percent require very special handling and shipping procedures. The kind used as a household disinfectant is typically only 3 percent hydrogen peroxide and 97 percent water.
Because high concentrations are hard to transport, and low concentrations, being mostly water, are uneconomical to ship, the material is often hard to get in places where it could be especially useful, such as remote communities with untreated water. (Bacteria in water supplies can be effectively controlled by adding hydrogen peroxide.) As a result, many research groups around the world have been pursuing approaches to developing some form of portable hydrogen peroxide production equipment.
Most of the hydrogen peroxide produced in the industrialized world is made in large chemical plants, where methane, or natural gas, is used to provide a source of hydrogen, which is then reacted with oxygen in a catalytic process under high heat. This process is energy-intensive and not easily scalable, requiring large equipment and a steady supply of methane, so it does not lend itself to smaller units or remote locations.
"There's a growing community interested in portable hydrogen peroxide," Surendranath says, "because of the appreciation that it would really meet a lot of needs, both on the industrial side as well as in terms of human health and sanitation."
Other processes developed so far for potentially portable systems have key limitations. For example, most catalysts that promote the formation of hydrogen peroxide from hydrogen and oxygen also make a lot of water, leading to low concentrations of the desired product. Also, processes that involve electrolysis, as this new process does, often have a hard time separating the produced hydrogen peroxide from the electrolyte material used in the process, again leading to low efficiency.
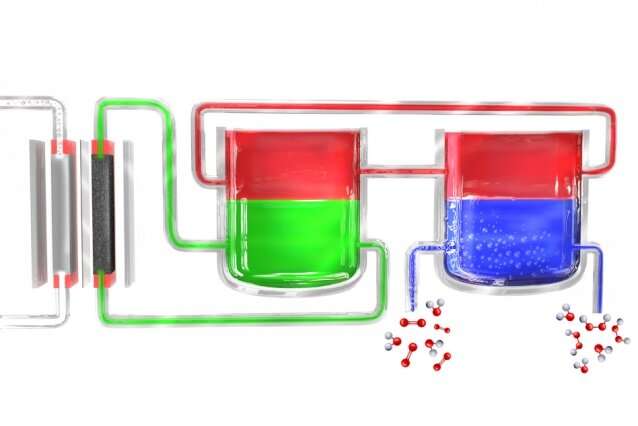
Surendranath and the rest of the team solved the problem by breaking the process down into two separate steps. First, electricity (ideally from solar cells or windmills) is used to break down water into hydrogen and oxygen, and the hydrogen then reacts with a "carrier" molecule. This molecule—a compound called anthroquinone, in these initial experiments—is then introduced into a separate reaction chamber where it meets with oxygen taken from the outside air, and a pair of hydrogen atoms binds to an oxygen molecule (O2) to form the hydrogen peroxide. In the process, the carrier molecule is restored to its original state and returns to carry out the cycle all over again, so none of this material is consumed.
The process could address numerous challenges, Surendranath says, by making clean water, first-aid care for wounds, and sterile food preparation surfaces more available in places where they are presently scarce or unavailable.
"Even at fairly low concentrations, you can use it to disinfect water of microbial contaminants and other pathogens," Surendranath says. And, he adds, "at higher concentrations, it can be used even to do what's called advanced oxidation," where in combination with UV light it can be used to decontaminate water of even strong industrial wastes, for example from mining operations or hydraulic fracking.
So, for example, a portable hydrogen peroxide plant might be set up adjacent to a fracking or mining site and used to clean up its effluent, then moved to another location once operations cease at the original site.
In this initial proof-of-concept unit, the concentration of hydrogen peroxide produced is still low, but further engineering of the system should lead to being able to produce more concentrated output, Surendranath says. "One of the ways to do that is to just increase the concentration of the mediator, and fortunately, our mediator has already been used in flow batteries at really high concentrations, so we think there's a route toward being able to increase those concentrations," he says.
"It's kind of an amazing process," he says, "because you take abundant things, water, air and electricity, that you can source locally, and you use it to make this important chemical that you can use to actually clean up the environment and for sanitation and water quality."
"The ability to create a hydrogen peroxide solution in water without electrolytes, salt, base, etc., all of which are intrinsic to other electrochemical processes, is noteworthy," says Shannon Stahl, a professor of chemistry at the University of Wisconsin, who was not involved in this work. Stahl adds that "Access to salt-free aqueous solutions of H2O2 has broad implications for practical applications."
Stahl says that "This work represents an innovative application of 'mediated electrolysis.' Mediated electrochemistry provides a means to merge conventional chemical processes with electrochemistry, and this is a particularly compelling demonstration of this concept. … There are many potential applications of this concept."
More information: "Electrosynthesis of hydrogen peroxide by phase-transfer catalysis." Joule (2019). DOI: 10.1016/j.joule.2019.09.019
Journal information: Joule
Provided by Massachusetts Institute of Technology
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.




















