Catalytic immunotherapy for cancer: Nanoparticles act as artificial enzymes
Theoretically, our immune system could detect and kill cancer cells. Unfortunately, tumors are well armed to fight these attacks. Despite modern cancer treatments, metastases and relapses remain a major problem. Increasing anti-tumor immunity might now be made possible, thanks to copper telluride nanoparticles that mimic enzymes, especially under NIR-II light irradiation. This induces oxidative stress in tumor cells, quashing their immunosuppressive status and triggering inflammatory processes, as Chinese scientists report in the journal Angewandte Chemie.
So-called nanoenzymes are hot candidates for treatments called catalytic immunotherapy. These nanoparticles have structures completely different from biological enzymes but mimic the enzymes' catalytic activity. They are easier to prepare than natural enzymes, as well as being less expensive and more stable.
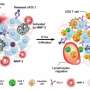
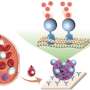
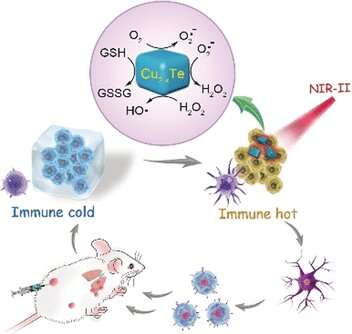
Researchers working with Wansong Chen and You-Nian Liu at Central South University (Changsha, Hunan, China) have now introduced a new nanoenzyme: copper telluride nanoparticles (Cu2-xTe) mimic the activities of the enzymes glutathione oxidase and peroxidase. The activity of this nanoenzyme is due to the copper ions, which switch between two oxidation states. The substrate is glutathione, an antioxidant that is found in significantly higher concentrations in tumor cells than in healthy ones. This is why the nanoenzymes are only active in tumor cells. In addition, they absorb NIR-II light (near-infrared light with a wavelength between 1000 and 1350 nm), causing their local environment to heat up. This effect strongly increases the enzyme-like activity of the nanoenzymes. In treatment, the NIR-II irradiation could be selectively applied to the tumor.
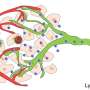
Experiments in tumor-cell cultures and with tumor-bearing mice demonstrated that use of the nanoenzymes and NIR-II irradiation triggers a whole cascade of cellular responses that greatly increase the oxidative stress within the tumor, which ultimately leads to cell death. At the same time, the suppression of immunological processes in the microenvironment around the tumor is lifted. Instead, substances that promote inflammation are released and an immune response is triggered. This is supported by the elevated concentration of effector T cells observed by the researchers. The immune system is thus able to learn to defend itself against metastasis and to build an "immunological memory" to prevent relapses.
More information: Mei Wen et al. Artificial Enzyme Catalyzed Cascade Reactions: Antitumor Immunotherapy Reinforced by NIR-II Light, Angewandte Chemie International Edition (2019). DOI: 10.1002/anie.201909729
Journal information: Angewandte Chemie , Angewandte Chemie International Edition
Provided by Wiley