Study reveals the intertwined response of pathogen and host during fungal infections
A study published in the journal Nature Communications reveals that cells of a fungal pathogen and cells from the host's immune system engage in a tightly coordinated dance of gene expression as the fungus seeks to launch an infection and the host endeavors to protect itself. This never before observed coordination may point the way to new tools for studying fungal diseases and provide new clues into how to treat fungal infections, including sometimes deadly systemic infections.
The study, "Coordinated host-pathogen transcriptional dynamics revealed using sorted subpopulations and single macrophages infected with Candida albicans," was conducted by a team that includes Reeta Rao, professor of biology and biotechnology at WPI and a visiting scientist at the Broad Institute of MIT and Harvard, and Toni Delorey, a Ph.D. candidate at WPI. Rao and Christine Cuomo, senior group leader at the Broad Institute, are the lead authors.
Fungal diseases, particularly those caused by Candida, a genus prevalent in humans that is responsible for oral thrush, ear infections, and vaginitis, are notoriously difficult to treat. With a 30 to 50 percent mortality rate, systemic Candida infections, which can occur in immunocompromised individuals, are among the leading causes of serious illnesses and death in hospitalized patients.
Because of the lackluster effect of most antifungal medications, scientists study the biology of Candida and its interaction with the mammalian immune system looking for potential vulnerabilities to exploit or possible targets for
new drugs. In the current study, Rao and the WPI/Broad Institute team focused on the interaction between a Candida species known as C. albicans, the most common fungal pathogen, and macrophages, immune cells that attack invaders by engulfing and digesting them.
In previous research, Rao and her colleagues found that when populations of C. albicans and macrophages are mixed, the outcomes of their interactions are dynamic and heterogenous. Microscopic observation reveals
that macrophages appear to consume and successfully destroy the fungus or consume the fungus but are lysed by fungi that are filamenting, allowing the fungus to escape. In addition, some subpopulations of macrophages are seemingly idle and not pursuing the fungi.
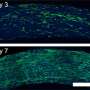
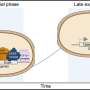
To understand the transcriptional wiring behind these diverse outcomes, the WPI/Broad Institute researchers developed a novel pipeline for studying the host-pathogen responses of individual fungus and macrophage cells over the course of an infection. The technique made it possible, for the first time, to observe and follow changes in gene expression in single host-pathogen pairs as they interact with one another.
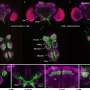
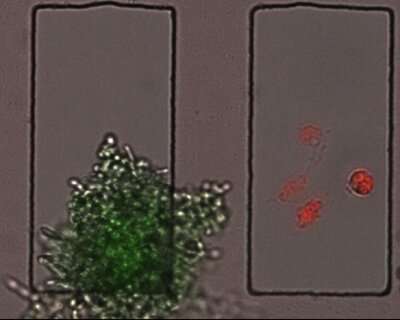
They did this by using different fluorescent dyes to mark the C. albicans cells and macrophages, and determined the state each cell type based on the appearance or disappearance of those colors. For example, C. albicans cells
were marked with both red and green fluorescent dyes. A living fungus inside a macrophage would emit both colors. But since the green dye cannot withstand the high pH inside phagosomes, the organelles within macrophages that digest captured microorganisms, dead C. albicans would emit only red light.
Using this tool, the researchers were able to parse four possible infection outcomes: a C. albicans engulfed and killed by a macrophage; a C. albicans engulfed but alive and probably filamenting; a C. albicans near a macrophage, but not engulfed; and a macrophage in the presence of a C. albicans but not engulfing it. They looked for these outcomes at two hours and at four hours after the start of an infection, and observed changes in gene expression in the two cell types.
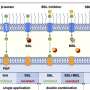
For example, at two hours, macrophages encountering fungus cells secreted chemicals called cytokines that promote inflammation and also recruit other macrophages. Inflammation helps the body respond to an infection. At this point most of the fungus cells were in the benign "yeast" form. The C. albicans engulfed by macrophages expressed genes that enable the fungus to survive inside the macrophage, where it is deprived of glucose, its normal food. Still, most of the fungal cells engulfed at this stage were likely to be killed. At four hours, C. albicans cells that had been engulfed by macrophages were expressing genes for cell wall remodeling and filamentation. In response, the macrophages began secreting a different kind of cytokine, one that serves to reduce inflammation.
"Inflammation is a good thing, initially," Rao said. "But prolonged inflammation is damaging to the body."
The researchers were especially interested in the population of macrophages that seemed to sit on the sidelines, remaining inactive as other macrophages around them are busy consuming C. albicans. Rao says both cell type (host and pathogen) seem to be following an evolutionary strategy that can best be described as hedging their bets—waiting to see the outcome of the macrophage vs. pathogen battle and standing ready to wade into the fight should the pathogen seem to be winning. In fact, both host and pathogen seem to engage in this strategy, the authors noted in the Nature Communications paper.
"Both cell types may rely on stochastic diversification of phenotypes to improve their survival rate in the event of an encounter with the other cell type," they wrote. "For instance, clonal populations of C. albicans that find themselves in the unpredictable, changing environment of a host phagocyte could increase the chance of survival by varying the expression of key genes involved in the response."
Rao noted that bet hedging has been observed in populations of pathogenic bacteria and cancer cells, and is associated with drug resistance. This is the first time it has been identified in a pathogenic fungus. She said that a better understanding of the intertwined fates of macrophages and C. albicans could help identify new strategies for treating fungal infections.
"Research like this could point to particular genes to target with drugs, but we know that penetrating the tough cell walls of fungi is difficult," she said. "A better strategy may be to use what we are learning about the evolution of an infection and about bet hedging to modulate the host immune system to make it more effective at combating the infection. For example, it might be possible to modulate the immune system by manipulating the signals that the macrophages send out to more effectively recruit a repertoire of immune cells to attack the invading fungi. I think this is where a lot of exciting new research is going to be done."
More information: José F. Muñoz et al. Coordinated host-pathogen transcriptional dynamics revealed using sorted subpopulations and single macrophages infected with Candida albicans, Nature Communications (2019). DOI: 10.1038/s41467-019-09599-8
Journal information: Nature Communications
Provided by Worcester Polytechnic Institute