August 21, 2019 report
Mimicking the diving bell spider to improve carbon conversion into fuels
A team of researchers affiliated with several institutions in France has found a way to improve the conversion of CO2 into fuels by mimicking the behavior of the diving bell spider. In their paper published in the journal Nature Materials, the group describes using captured air bubbles to improve the conversion efficiency of carbon dioxide into usable fuels.
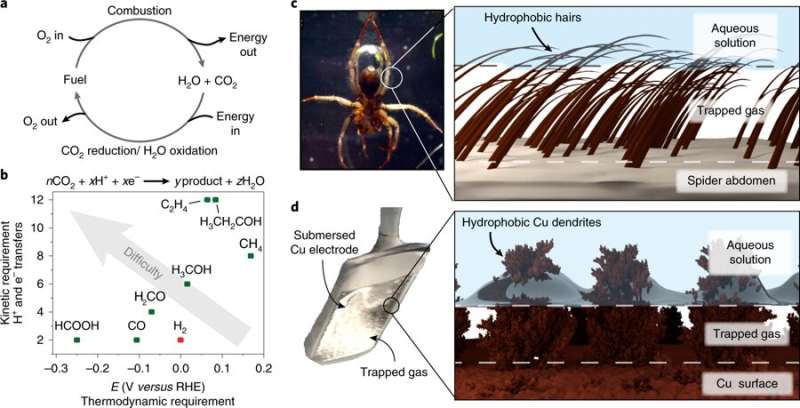
Current electrochemical processes that convert CO2 into hydrocarbons typically use copper as an electrocatalyst—it is generally coated with electrodes and immersed in a liquid that contains carbon dioxide. Applying electricity sets off the reduction process that converts the CO2 into methane, ethanol, ethylene and carbon dioxide. Unfortunately, such processes also produce hydrogen gas, which reduces the efficiency of the process. Efforts to improve the process have involved forming the electrodes into nanostructures or by doping the copper with other materials. But thus far, such efforts have not resulted in adequate efficiency improvements. In this new effort, the researchers looked to the diving spell spider for inspiration.
Diving bell spiders are able to swim underwater because they have strongly hydrophobic hairs on their underbelly that allow for trapping an air bubble, which the spider uses to breathe under water. The researchers thought that if the copper in a CO2 conversion process did roughly the same thing, more carbon dioxide would be exposed to the copper during conversion, improving efficiency. To that end, they forged a piece of copper with tiny, tree-like shapes on its surface and coated it with a hydrophobic material. When the copper was dunked into a CO2-containing solution, bubbles formed on the surface of the copper. And when electricity was applied, the conversion process occurred as usual, with one major difference. The process was much more efficient.
The researchers report that the efficiency of the conversion was approximately 56 and 17 percent for ethylene and ethanol, compared to 9 and 4 percent in conventional systems. Also, hydrogen production was measured at 10 percent, compared to 71 percent for traditional systems. The researchers note that more work is required, however, because the process requires more electricity than conventional systems.
More information: David Wakerley et al. Bio-inspired hydrophobicity promotes CO2 reduction on a Cu surface, Nature Materials (2019). DOI: 10.1038/s41563-019-0445-x
Journal information: Nature Materials
© 2019 Science X Network