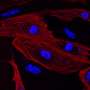
Transformer cells: Shaping cellular 'behaviour'
Scientists from Sechenov University, conjointly with their fellow Chinese and American researchers, have examined the latest advances in the use of skeletal muscle progenitor cells, specifying the core challenges inherent to the applicability of MPCs in cell therapy, and outlining the most promising breakthrough technologies. The outcomes of this research were reported in Applied Physics Reviews, the article having been roundly praised by the editorial board.
Progenitor cells are cells that have the capacity to evolve (or differentiate) into a specific type of cell, for instance, muscle tissue cells. This ability makes them key candidates for cell therapy in the treatment of damaged muscle tissue due to injury, disease, or age-associated dysfunctions. The technique could be described as follows: progenitor cells are harvested from the patient's healthy muscle tissue sample, cultivated in vitro and then grafted onto the patient's damaged tissues. The method requires the appropriate environment (similar to that in the human body) to enable the differentiation of progenitor cells under laboratory conditions. However, being highly sensitive to the subtlest changes in the growth-supporting microenvironment, progenitor cells may alter their behavioural patterns ex vivo and lose the ability to differentiate into target types of cells.
The research demonstrates that proper management of progenitor cell behaviour requires both a suitable scaffold (or a 'backbone' on which the tissue is cultivated) and extracellular matrix that interconnects the surrounding cells and regulates the intracellular processes.
Extracellular matrix that provides the microenvironment for progenitor cells in vivo contains hundreds of various proteins, lipids, and carbohydrates, which play a crucial role in tissue regeneration. This microenvironment is extremely active and its internal processes are essential for cell growth and migration. Despite the existing multitude of artificial extracellular matrices, including those derived from animal tissues, native human tissues remain the most favourable environment for cell cultivation.
Prior to publishing their report, the authors had designed extracellular matrix-derived scaffolds for biofabricating skin, skeletal muscle and kidney tissues that demonstrated excellent viability results due to their tissue-specific differentiation. In order to engineer functional matrices, any cells and their components that may trigger immune reaction during grafting are mechanically isolated, or washed out with processing solution, from the target tissue sample. The scientists have designed and tested a tissue decellularisation method that efficiently removes the cell components, while preserving its structural support—the matrix—and active compounds (cytokines, growth factors), which essentially control the cell behaviour. This was made possible by accelerating the decellularization process: the solution remains in contact with critical compounds for a shorter period of time, ensuring their integrity and viability. There also exist a number of extracellular matrix hydrogel types that have proven to be reasonably effective in tissue construction and nutrient supply.
As Peter Timashev, a contributing author and Director of the Institute for Regenerative Medicine of Sechenov University, remarked, "When engineering tissues or body organs in vitro, we always aim to create the sort of environment that would be as identical to the human body as practically possible. That being said, the sheer complexity of extracellular matrix makeup makes the fabrication of fully sustainable artificial matrices unachievable at this point in time. Therefore, our goal is to try to extract the matrix very carefully and use it in engineering target tissues—this technique will enable an accurate reproduction of living tissues in the future and facilitate their application in clinical settings."
More information: Longkun Li et al, Biofabrication of tissue-specific extracellular matrix proteins to enhance the expansion and differentiation of skeletal muscle progenitor cells, Applied Physics Reviews (2019). DOI: 10.1063/1.5088726
Provided by Sechenov University