A 'crisper' method for gene editing in fungi

CRISPR/Cas9 is now a household name associated with genetic engineering studies. Through cutting-edge research described in their paper published in Scientific Reports, a team of researchers from Tokyo University of Science, Meiji University, and Tokyo University of Agriculture and Technology, led by Dr. Takayuki Arazoe and Prof Shigeru Kuwata, has recently established a series of novel strategies to increase the efficiency of targeted gene disruption and new gene "introduction" using the CRISPR/Cas9 system in the rice blast fungus Pyricularia (Magnaporthe) oryzae. These strategies include quicker (single-step) gene introduction, use of small homologous sequences, and bypassing of certain prerequisite host DNA "patterns" and host component modification.
The team led by Dr. Arazoe and Prof Kuwata has devised quick, simple techniques for gene editing (target gene disruption, sequence substitution, and re-introduction of desired genes) using CRISPR/Cas9 in the rice blast fungus Pyricularia (Magnaporthe) oryzae, a type of filamentous fungus. Spurred on by encouraging results, the researchers surmise, "Plants and their pathogens are still coevolving in nature. Exploiting the mutation mechanisms of model pathogenic fungi as a genome editing technique might lead to the development of further novel techniques in genetic engineering."
The working component of the CRISPR/Cas9 system binds to the target gene region (DNA) and causes a site-specific double-stranded break (DSB) in the DNA. Effective binding of this component requires a certain "motif" or "pattern" called the protospacer-adjacent motif (PAM), which follows downstream of the target gene region.
Most genome editing techniques require DSBs induced at the target site, which trigger DNA "repair" pathways in the host. Homologous recombination (HR) is a mechanism for repair of DSBs, and it is useful because it adds complementary sequences. However, the underlying methodology is laborious, and its efficiency conventionally depends on external factors such as the host properties as well as PAMs. HR can be divided into two pathways: "noncrossover" (gene conversion) and "crossover" type. Crossover-type repairs are known to occur in cells that undergo meiosis. However, the understanding of their role in cells that undergo mitosis is limited, and such information on filamentous fungi is virtually unavailable. It is this gap in knowledge that the researchers were looking to address.
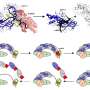
In their study, the researchers first created a vector (gene delivery system) based on CRISPR/Cas9 to confirm crossover-type HR in the recipient gene region in the rice blast fungus.
Then, to check gene targeting or "sequence substitution," they created a "mutant" vector, optimized for single crossover-type HR, for targeted disruption of the host gene that encodes scytalone dehydratase (SDH), a protein involved in melanin formation. This vector was introduced into the vector containing the gene for hygromycin B phosphotransferase (hph), which confers resistance to the antibiotic hygromycin B. The researchers speculated that the single crossover-type HR would insert the entire vector along with hph into the target site. The mutants with disrupted SDH gene would be identified as white colonies (owing to loss of melanin) on a medium containing hygromycin B. The researchers found that the number of hygromycin B-resistant white colonies dramatically increased by using the CRISPR/Cas9 vector, which means that the CRISPR/Cas9 system is effective in inducing single crossover-type HR. The greatest benefit of this technique is that it needs extremely short homologous sequences (100 base pairs; which is really small in molecular biology).
The researchers also used a similar strategy to check whether gene introduction (or "knock in") is possible via single crossover-type HR using a CRISPR/Cas9 vector. They used the green-fluorescent protein (GFP) gene, which is widely used as a "reporter" gene to make host cells glow fluorescent green when inserted into their genome. They speculated that single crossover HR would result in introduction of GFP into the recipient sequence. Indeed, they found that use of the CRISPR/Cas9 vector gave rise to green fluorescent colonies on hygromycin medium. These findings show that the CRISPR/Cas9 system can be used for efficient "one-step" gene knock-in.
This research points towards a surprising fact that, perhaps, PAMs are not all that necessary for CRISPR/Cas9 gene editing in fungi. Hailing the success of the research, the team states, "We have found that filamentous fungi have unique genomic characteristics, wherein crossovers are frequently induced, even in somatic cells, by cleaving the target DNA. We used these characteristics to disrupt the target DNA and to introduce "reporter" genes. We also succeeded in increasing the efficiency and speed of the knock-in, using a single-step process. This technology overcomes the restriction posed by PAMs—which is one of the biggest disadvantages of the CRISPR/Cas9 system—and enables more flexible genome editing, which has been difficult in previous studies on filamentous fungi."
Finally, when asked about the broader applications of this research, Dr. Arazoe and Prof Kuwata eloquently state, "Rice blast fungus is an important pathogen that causes destructive disease of rice, which is the staple food of the country. The CRISPR/Cas9-based genome editing technique developed in our study can speed up molecular biological research on this pathogen, ultimately contributing to stable food supply and plant-based food safety. Also, this technique is applicable to other filamentous fungi widely used in industry—especially in the bioprocessing, food, and fermentation industries."
More information: Tohru Yamato et al, Single crossover-mediated targeted nucleotide substitution and knock-in strategies with CRISPR/Cas9 system in the rice blast fungus, Scientific Reports (2019). DOI: 10.1038/s41598-019-43913-0
Journal information: Scientific Reports
Provided by Tokyo University of Science