May 21, 2019 report
Amyloid fibrils lit with near-infrared radiation found to emit a dim, near-infrared signal
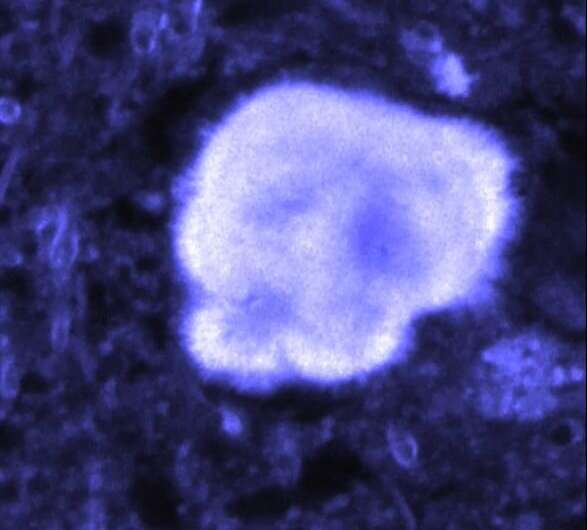
A team of researchers affiliated with several institutions in France has found that amyloid fibrils lit with near-infrared radiation emit a dim, near-infrared signal. In their paper published in the journal Nature Photonics, the group describes their study of amyloid fibrils and plaques in mice and humans and what they found.
Amyloid fibrils are tiny structures that self-form in some proteins. When they clump together, they form what are known as amyloid plaques. They are associated with the development of neurological diseases such as Alzheimer's and Parkinson's disease. Despite years of study, it is still not known what causes them. In this new effort, the researchers sought to learn more about the early stages of fibril development by developing a way to see it happening.
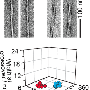
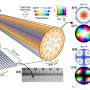
Prior work had shown that when ultraviolet light shines onto tissue-containing proteins, the tissue emits blue light. Researchers have found that the emissions become stronger if there are fibrils present in the proteins. While this finding has been useful, it has only allowed for superficial study of fibril formation due to the shallow depth of UV and blue light penetration. In their experiments, the researchers tried firing near-field radiation at sample human proteins and found that and fibrils present would emit a dim, near-infrared signal. This was important, because unlike UV light, near-field radiation can penetrate relatively deeply into tissue.
Next, the researchers genetically developed an Alzheimer's mouse model, opened their skulls and fired near-field radiation at living brain tissue, observing a near-infrared signal. They suggest that in the future, it might be possible to use their technique to test for Alzheimer's disease in humans—currently the only way to do so is by using cognitive tests. How such a test might work, though, is not clear. With present technology, such a test would involve opening the skull and inserting a probe—an option most patients would not choose. The researchers acknowledge such roadblocks, but suggest that in the future, new technology could allow such a test without surgery. They report that the unique properties of amyloid fibrils could lead to new biophotonic devices.
More information: Jonathan Pansieri et al. Ultraviolet–visible–near-infrared optical properties of amyloid fibrils shed light on amyloidogenesis, Nature Photonics (2019). DOI: 10.1038/s41566-019-0422-6
Journal information: Nature Photonics
© 2019 Science X Network