Heterogeneous catalyst goes enzymatic
What if there were no tunnels in the Swiss Alps? Anyone trying to travel through them would have to go up and down hills and zigzag around the ranges. A lot more energy and time is saved by passing through a tunnel than climbing a mountain. This is similar to how catalysts work: they speed up chemical reactions by lowering the energy required to reach the desired physical state.
In industrial manufacturing processes, heterogeneous catalysis, which typically involves the use of solid catalysts placed in a liquid or gas reaction mixture, has many potential applications. Being in a different phase, heterogeneous catalysts can be easily separated from a reaction mixture. In this way, the catalysts can be effectively recovered and recycled, being quite eco-friendly. In addition, they exhibit very stable activity even under harsh reaction conditions. Despite such advantages, heterogeneous catalysis has been thought to allow for less interaction and controllability than homogeneous catalysis due to a poor understanding of its reaction process.
Researchers at the Center for Nanoparticle Research (led by Director Taeghwan Hyeon) within the Institute for Basic Science (IBS) in collaboration with Professor Ki Tae Nam at Seoul National University and Professor Hyungjun Kim at KAIST demonstrated enzyme-like heterogeneous catalysis for the first time. They developed a highly active heterogeneous TiO2 photocatalyst incorporated with many single copper atoms. They used this catalyst for photocatalytic hydrogen production, and found that the catalyst is as active as the most active and expensive Pt-TiO2 catalyst.
The researchers were committed to modeling the catalyst structure in a similar way to the most efficient and reactive catalysts that are biological enzymes. Enzymes comprise catalytically active metal atoms and surrounding proteins that work very closely to keep their feedbacks moving back and forth. Thanks to this cooperative internal communication, enzymes can swiftly adapt their structure to optimally fit for desired reactions (generally known as the induced-fit model). During the adaptation, enzymes intermittently return to their original shapes and turn reformed. Professor Hyeon says, "For the first time, we found that an enzyme-like reversible and cooperative activation process occurs even in heterogeneous catalysts. This is an unprecedented platform that merges advantages of both heterogeneous catalysts and biological enzymes. While featuring the robust stability of heterogeneous catalysts, cooperative and reversible characteristics of enzymes adds significant controllability, which in the end brings high activity for hydrogen (the most efficient and ideal fuel) production from photocatalytic water splitting reactions."
Biological enzymes have been considered as a central model for developing artificial catalysts. They have been successfully utilized in designing homogeneous catalysts for various reactions. Still, there had been no report on industrially important heterogeneous catalysts with these enzyme-like characteristics due to the lack of atomic-level understanding of heterogeneous catalysts. This new study demonstrates that heterogeneous catalysts can work like enzymes, confirming the fundamental principle that cooperative interplay between atomic catalysts and adjacent environments have significant influence on overall material properties and catalytic activity.
Combining theoretical simulations and nanomaterial synthesis technologies, the researchers synthesized an enzyme-like heterogeneous catalyst. (Fig. 2) They covered a round-shaped TiO2 substrate with single-atom copper. They wrapped TiO2 and copper atoms together. Subsequent baking successfully stabilized copper single atoms exclusively on titanium sites. It was crucial for this study to design site-specific single atom catalysts, since this single-atom structure directly mimics the structure of enzymes (made of single atom metallic ions and surrounding proteins).
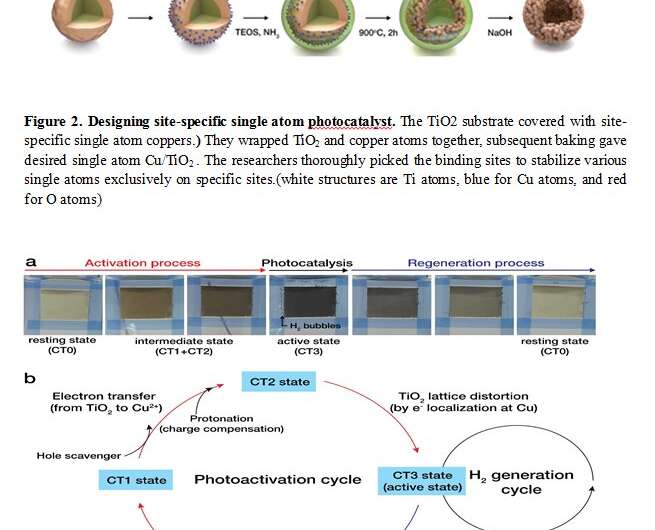
Interestingly, the synthesized site-specific single atom Cu/TiO2 catalysts underwent a unique photoactivation process. By absorbing light, TiO2 excites an electron. The excited electron is transferred to a single copper atom through a simple oxidation state change. The transfer of an electron in turn changes back surrounding TiO2 structures (just like the induced-fit model of an enzyme). This active state then reverses to the initial resting state as an electron is transferred back to the TiO2 from a metal atom. In fact, this interactive and reversible mechanism was confirmed with the white Cu/TiO2 rapidly turning to black under light irradiation, and back to its initial white color when purged with air. Thanks to these enzymatic characteristics, single atom Cu/TiO2 catalyst turned over 40% of light energy into H2, an exceptionally high catalytic activity, which is as active as the most active and expensive Pt-TiO2 photocatalyst. Hydrogen is known to be the most efficient and ideal fuel because it generates water as the only byproduct.
More information: Reversible and cooperative photoactivation of single-atom Cu/TiO2 photocatalysts, Nature Materials, DOI: 10.1038/s41563-019-0344-1 , www.nature.com/articles/s41563-019-0344-1
Journal information: Nature Materials
Provided by Institute for Basic Science