A new catalyst for water splitting that is the best of both worlds
Taking water and ripping it apart into hydrogen and oxygen could form the basis of artificial photosynthetic devices that could ultimately power homes and businesses. However, catalysts, including those used to "split" water, have either worked well but are expensive and unstable, or are affordable and stable, but don't work as well. Now, researchers report in ACS Central Science a new catalyst that is really the best of both worlds.
Identifying ideal materials that can split water is a long-standing problem in renewable energy storage. Catalysts, which help reactions occur, are often used in this process. "Homogeneous" ones dissolve into the reaction solution and are usually active and selective. However, they don't work well in some applications because they are unstable and expensive. In contrast, "heterogeneous" catalysts are solids that are stable, recyclable and convenient to work with, but they are usually not very active or selective. Dunwei Wang and colleagues proposed they could get closer to the ideal catalyst by producing a hybrid material.
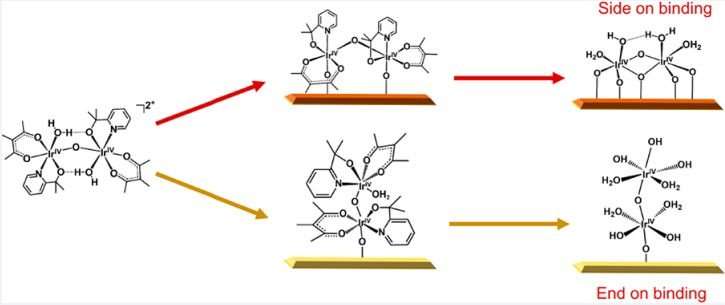
The researchers developed a new hybrid catalyst made of iridium dinuclear heterogeneous catalysts (DHCs) attached to a tungsten oxide substrate. They found that attaching the ends of the DHC molecules—instead of the sides—allowed the catalyst to perform optimally. The researchers suggest that this first-of-its-kind material could be an important step toward alternative solar energy storage or artificial photosynthesis.
More information: "End-On Bound Iridium Dinuclear Heterogeneous Catalysts on WO3 for Solar Water Oxidation" pubs.acs.org/doi/full/10.1021/acscentsci.8b00335
Abstract
Heterogeneous catalysts with atomically defined active centers hold great promise for high-performance applications. Among them, catalysts featuring active moieties with more than one metal atom are important for chemical reactions that require synergistic effects but are rarer than single atom catalysts (SACs). The difficulty in synthesizing such catalysts has been a key challenge. Recent progress in preparing dinuclear heterogeneous catalysts (DHCs) from homogeneous molecular precursors has provided an effective route to address this challenge. Nevertheless, only side-on bound DHCs, where both metal atoms are affixed to the supporting substrate, have been reported. The competing end-on binding mode, where only one metal atom is attached to the substrate and the other metal atom is dangling, has been missing. Here, we report the first observation that end-on binding is indeed possible for Ir DHCs supported on WO3. Unambiguous evidence supporting the binding mode was obtained by in situ diffuse reflectance infrared Fourier transform spectroscopy and high-angle annular dark-field scanning transmission electron microscopy. Density functional theory calculations provide additional support for the binding mode, as well as insights into how end-on bound DHCs may be beneficial for solar water oxidation reactions. The results have important implications for future studies of highly effective heterogeneous catalysts for complex chemical reactions.
Journal information: ACS Central Science
Provided by American Chemical Society