Keeping genetic engineering localized
Genetic engineering tools that spread genes within a target species have the potential to humanely control harmful pests as well as eradicate parasitic diseases such as malaria.
The tools, known as gene drives, ensure that engineered organisms transmit desired genetic variants to their offspring. These variants could ensure, for example, that the organisms only produce male offspring, or sterile females.
In this way, gene drives could be used to exterminate insects such as mosquitoes that carry pathogens, and that can spread malaria, dengue, and the Zika virus. Gene drives could also be used to control invasive species such as rodents that can threaten the survival of native animals.
However, previously described versions of gene drives based on the CRISPR genome editing system have the potential to spread far wider than their intended local population—to affect an entire species. The affects could also spread across international boundaries, potentially leading to disputes between countries where no prior agreement had been made.
These types of concerns could significantly delay, if not altogether prevent, the safe testing and introduction of the technology.
Now, in a paper published today in the Proceedings of the National Academy of Sciences, researchers at MIT and Harvard University describe a gene drive system with in-built controls.
The CRISPR-based drive consists of a series of genetic elements arranged in a so-called daisy chain, according to Kevin Esvelt, an assistant professor of media arts and sciences and head of the Sculpting Evolution research group at the MIT Media Lab who co-led the research.
One link within the daisy-drive system encodes the CRISPR gene editing system itself, while each of the other links encode guide RNA sequences. These guide sequences tell the CRISPR system to cut and copy the next link in the chain, Esvelt says.
Adding more links allows the daisy drive system to spread for additional generations within the population.
"Imagine you have a chain of daisies, and at each generation you remove the one on the end. When you run out, the daisy chain drive stops," Esvelt explains.
In this way, a small number of genetically-engineered organisms could be released into the wild to spread the daisy-drive within the local population, and then stop when programmed to.
"We're programming the organism to do CRISPR genome editing on its own, within its reproductive cells, in each generation," Esvelt says.
-
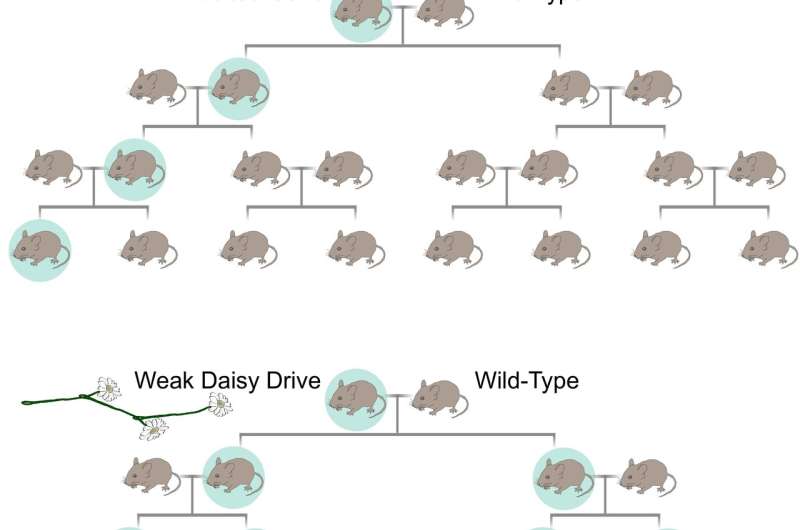
Unlike a self-propagating gene drive, a daisy drive system is predicted to be capable of editing a single population of wild organisms without spreading to all connected populations. Credit: K. Esvelt/MIT Media Lab -
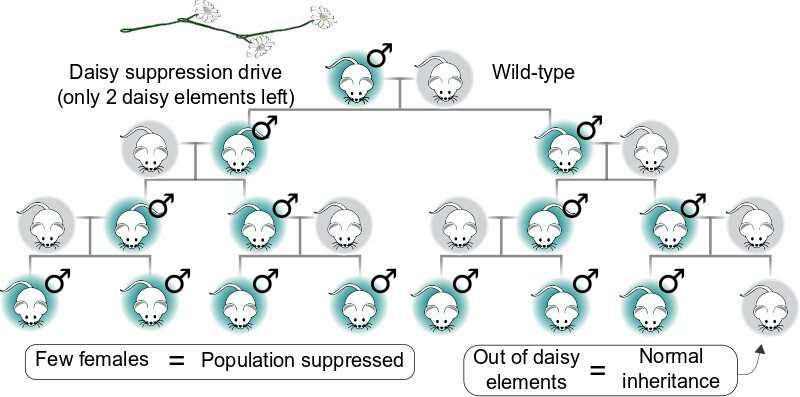
A daisy drive might be used to suppress populations by ensuring the preferential inheritance of a male-determining element. Because it eventually runs out of daisy links and stops, it will only affect local populations. Credit: K. Esvelt/MIT Media Lab -
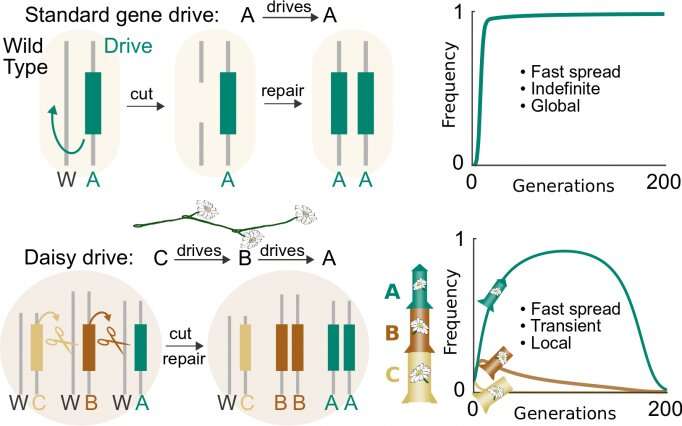
A standard CRISPR-based gene-drive system cuts the wild-type chromosome in the reproductive cells of each generation, causing its own DNA to be copied and ensuring that it will be inherited. Because it has everything it needs to favor itself, it can spread indefinitely through all connected populations. In contrast, a daisy-chain drive system has several components linked such that each copies the next in the chain. The one on the end has no inheritance advantage and can be lost; once gone, the next loses its advantage, and so on until all copies are gone and the drive stops. Credit: K. Esvelt/MIT Media Lab
Esvelt developed the system in collaboration with George Church, a professor of genetics at Harvard Medical School, visiting professor at the Media Lab, and a senior associate member at the Broad Institute of MIT and Harvard. Co-first authors Charleston Noble and John Min, both graduate students at Harvard Medical School, led the modelling and the molecular biology experiments designed to ensure the system is evolutionarily stable, respectively.
"If the world is to benefit from new gene-drive technologies, we need to be very confident that we can reverse it and contain it, both theoretically and via controlled tests," Church says.
"Many of the applications of gene drives involve islands and other geographical isolations, at least for initial tests, including invasive species and Lyme disease," he noted. "It would be great if these highly motivated local governments can do tests that do not automatically affect adjacent islands or mainlands. The daisy-chain drives offer this."
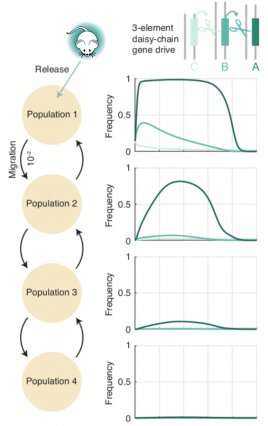
The research suggests that for every 100 wild counterpart, releasing just one engineered organism with a weak 3-link daisy-drive system, once per generation, should be enough to edit the entire population in about two generations—roughly a year in a fast-reproducing insect. That compares with existing systems that must release at least as many organisms as are already present in a local population, and sometimes 10 or 100 times as many.
The process could take several years in species that reproduce more slowly, such as mice, but would be more humane than the existing use of rodenticides, which can also harm people and predator species, Esvelt says.
In 2014, Esvelt and his colleagues first suggested that CRISPR-Cas9 could be used in gene-drive systems, and he has felt a moral responsibility to develop an alternative to self-propagating systems, he says. "Ideally, localization will let each community make decisions about its own environment, without forcing those decsions on others.
According to Professor Luke Alphey, head of arthropod genetics at The Pirbright Institute in the UK, self-propagating drive systems can spread rapidly through target populations. However, such drive systems are also thought likely to spread to all connected populations of the target species—which is desirable if you want to modify the entire species, undesirable if you do not, he says.
"Daisy-drives potentially provide a means to get much of the benefit of this type of gene drive, while constraining spread and also limiting persistence of the gene drive even in the target population," Alphey says. "That is likely to be highly desirable when one wants to affect one population but not another of the same species, perhaps affecting an invasive pest population but not populations of the same species in its native range."
Alphey was not involved in the initial daisy-drive research, but is now collaborating with Esvelt, including work on the use of daisy-drives in mosquitoes.
Esvelt and the Sculpting Evolution group are also beginning to explore the possible use of this technology to heritably immunize white-footed mice, the primary reservoir of the bacteria responsible for Lyme disease in North America. They are also setting up a research collaboration to explore the use of daisy-drives in Cochliomyia, also known as the New World screwworm, a parasitic fly that produces larvae that eat the living tissue of warm-blooded animals, causing considerable suffering.
In addtition, the researchers are also investigating this technology for use in nematode worms, microscopic creatures that reproduce every three days. This will allow them to carry out laboratory-based evolutionary studies of the daisy-drive engineered organisms, with the goal of ensuring the systems cannot become self-propagating.
More information: Charleston Noble et al. Daisy-chain gene drives for the alteration of local populations, Proceedings of the National Academy of Sciences (2019). DOI: 10.1073/pnas.1716358116
Journal information: Proceedings of the National Academy of Sciences
Provided by Massachusetts Institute of Technology
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.