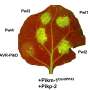
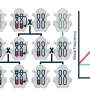
Gene drives have the potential to suppress mosquito populations, but resistant mosquitoes crop up

Researchers successfully built a gene drive to reduce female fertility in the mosquito that spreads malaria, but mutations gradually arose that blocked the spread of the new genes. Tony Nolan of Imperial College London, UK, and colleagues report these findings in a new paper in PLOS Genetics.
Gene drives have incredible potential for controlling insects that carry disease or destroy crops, by altering genes in ways that reduce population size or prevent the insect from spreading a parasite, virus or bacterium. In synthetic gene drives, scientists engineer genes that will quickly spread through a population because they are preferentially inherited by the offspring, even if they have a negative impact on the insect. Nolan and colleagues previously generated a synthetic gene drive in captive Anopheles gambiae mosquitoes, targeting genes involved in egg production to reduce the number of offspring.
They used the gene editing technology CRISPR/Cas9 to copy the gene drive onto both chromosomes during sperm and egg formation, so that the majority of offspring would inherit the engineered genetic trait. In just four generations, the new genes had spread through the mosquito population. As the gene drive continued, however, mutations gradually arose that blocked the engineered genes from being copied, which restored the females' fertility.
The study is the first to document the rise of mutations that make mosquitoes resistant to a gene drive, due to natural selection. These findings will allow researchers to make better predictions of how a gene drive will proceed and to improve the design of future gene drives to decrease the likelihood of resistance.
Tony Nolan adds: "Reducing the numbers of mosquito vectors has been the most effective tool to date for the control of malaria, so self-sustaining gene drives designed with this purpose have great potential. However gene drives are not a silver bullet and just like antibiotics can select for resistance in bacteria, gene drives can be susceptible to resistance at their target site. The novelty of this study is not that resistance emerges - we have been planning strategies to deal with this from the start - but that it documents the way it emerges and the way it is selected over generations. This work will help a lot in planning for and managing the emergence of resistance."
More information: Andrew M. Hammond et al, The creation and selection of mutations resistant to a gene drive over multiple generations in the malaria mosquito, PLOS Genetics (2017). DOI: 10.1371/journal.pgen.1007039
Journal information: PLoS Genetics
Provided by Public Library of Science