Study identifies a 'sensor' that activates cell migration
The cytoskeleton is a structure that not only helps cells maintain their shape and internal organisation, but also enables them to perform functions like movement and migration to sites far from the place where they originated. Migration is an essential part of the spread of cancer cells to another organ or tissue (metastasis).
To date, this association is not fully understood. How is the mechanical force generated by the cytoskeleton transformed into a biochemical signal that controls cell migration? During his work at the Spanish National Cancer Research Centre (CNIO), Daniel Lietha led an international study to answer this question. The resulting study, published in Proceedings of the National Academy of Sciences (PNAS), finds that the protein FAK is a key molecule that responds to the forces generated by the cytoskeleton, activating biochemical signals regulating cell adherence and migration. These findings broaden the knowledge of tumor invasion and metastasis.
Focal adhesion kinase (FAK) is present in the cytoskeleton and anchors the cell to the components of its external surroundings. "It is a signaling molecule that is key in a structure activated by forces that are generated by the contraction of the actin filaments in the cytoskeleton," explains Lietha, co-corresponding author of the study. "Until now, it was unclear whether FAK can act as a sensor for these forces required for cell movement. Based on the hypothesis that FAK is such a sensor, we carried out our study."
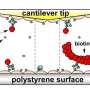
The researchers used atomic force spectroscopy to simulate the forces generated by the contraction of actin filaments in the cytoskeleton. They found that FAK changed as a response to these forces. One of these changes was related to the shape that FAK adopts when it activates the biochemical signaling involved in cell migration.
Atomic force spectroscopy is a technology to study the behaviour of specimens under force including intact cells and single molecules. "Actually, we have used a specific configuration of this technology pioneered by Hermann Gaub's team at the LMU, whose sensitivity and precision provide measurements of a single molecule with a high degree of accuracy," says Lietha.
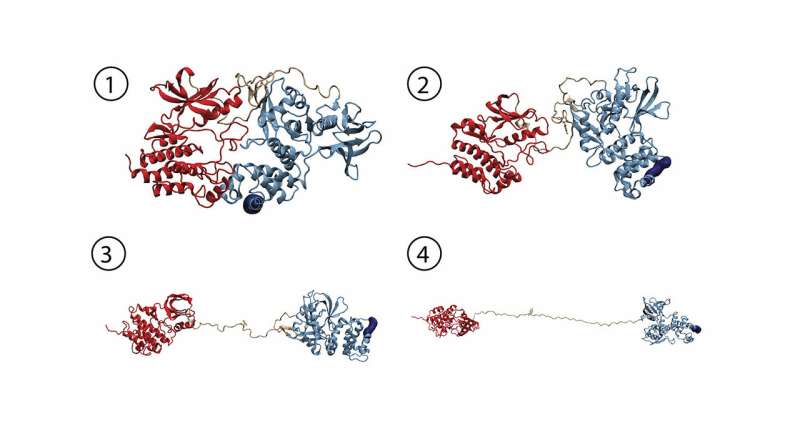
Thanks to this technology, the researchers obtained detailed mechanical information on FAK, which, together with knowledge of its atomic structure, can be interpreted in terms of how force induces changes in the structure to activate FAK. The Molecular Biomechanics group at HITS simulated this process on high-performance computers to obtain a fully dynamic view of these events. "We successfully uncovered key steps in FAK activation in great detail," says group leader Frauke Gräter. "We could show that this signaling protein not only gets activated by force, but also stays active while being further stretched out."
The mechano-environment of a cell influences its behavior. The focal adhesion kinase acts as sensor for changes in the extracellular and intracellular framework. "In single-molecule atomic force microscopy experiments and steered molecular dynamics simulations, we observed a force-mediated opening of the auto-inhibitory complex of FAK, and consequently, the activation of the kinase," says Magnus Bauer, first author of the publication and graduate student in the group of Hermann Gaub at the LMU, the experimental approach in Munich.
In cells, the mechano-activation of FAK is triggered upon stress between the cytoskeleton and the extracellular matrix. "With FAK, it seems we found the first non-muscle enzyme to be directly activated by mechanical force," says Bauer. "Transferring our data into cellular systems, we assume that translation of physiological forces into the biochemical signals could be one way to trigger the migration of cancer cells."
These are key findings to go deeper into the study of tumor invasion and metastasis. "In tumors, stiffening of the stroma results in increased forces that activate signaling pathways via FAK, that strongly promote tumor invasion and metastasis," says Lietha. The stroma is a tissue that surrounds cancer cells in tumors and affects their progression. Understanding how stroma stiffening and its implication in the activation of adhesion signaling are related to tumor invasion can provide the basis for the development of therapies targeting this mechanism.
Now that it has been found that the forces generated by the cytoskeleton prompt conformational changes in FAK, further studies are needed to measure the biochemical signals in response to mechanical forces.
More information: Magnus Sebastian Bauer el al., "Structural and mechanistic insights into mechanoactivation of focal adhesion kinase," PNAS (2019). www.pnas.org/cgi/doi/10.1073/pnas.1820567116
Journal information: Proceedings of the National Academy of Sciences
Provided by The National Centre for Cancer Research