March 21, 2019 feature
Optical force-induced self-guiding light in human red blood cell suspensions
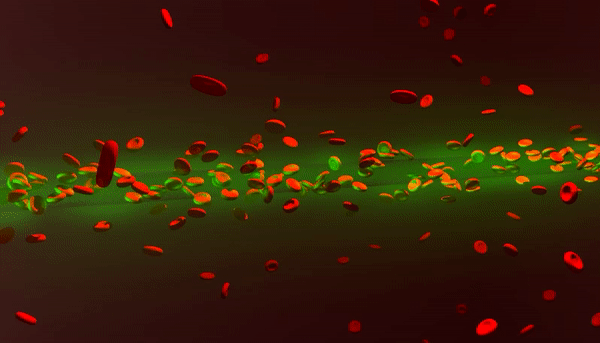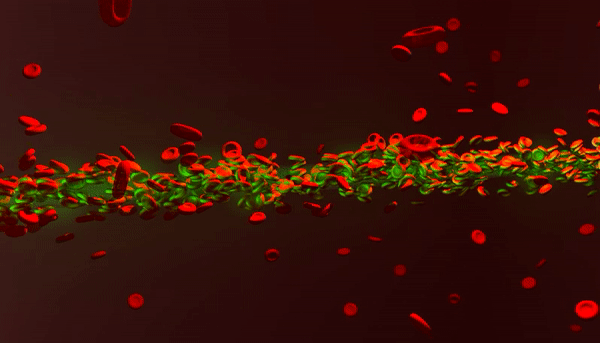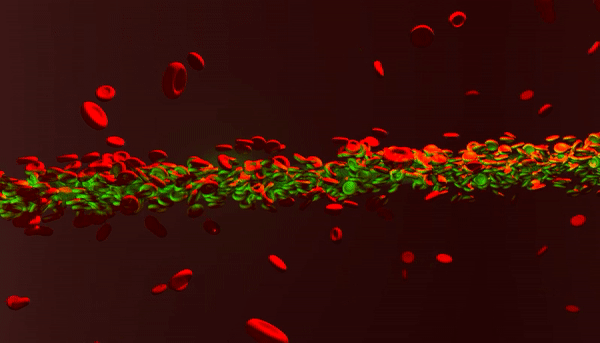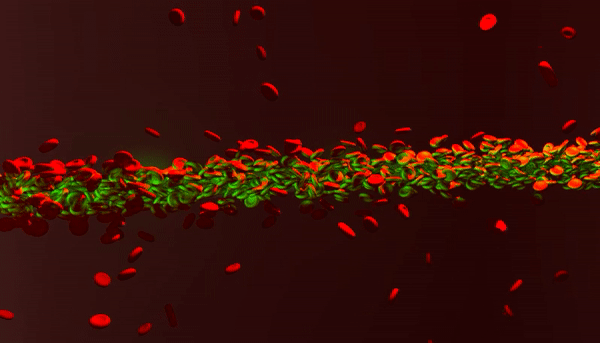
New photonic tools for medical imaging can be used to understand the nonlinear behavior of laser light in human blood for theranostic applications. When light enters biological fluids it is quickly scattered, however, some cell suspensions can induce nonlinear responses in laser beams to self-focus and enhance the penetration of light for biomedical applications as a quantifiable marker of disease. In a recent study now published in Light: Science and Applications, Rekha Gautam and her colleagues at the San Francisco State University and an international team of co-workers showed that a laser beam shining through red blood cell suspensions could become "self-trapped." The process reduced light scattering to retain the power of the beam of laser light within the biological samples.
The observed nonlinearity depended on osmotic conditions and the age of the samples. The scientists propose using the technique to diagnose sickle cell anemia or malaria; diseases which impact the size and shape of blood cells. Osmotic conditions play an important role in the properties of human red blood cells (RBCs) crucial during disease analysis. Numerous efforts in the past decade have focused on the study of the biomechanical properties of RBCs suspended in varying osmotic solutions.
In the present work, Gautam et al. determined the self-trapping and scattering-resistant nonlinear propagation of a laser beam through three different osmotic solutions/conditions. The results showed that the strength of the optical nonlinearity increased with osmotic pressure on the cells. Interestingly, in aged blood samples with lysed cells the nonlinear behavior was notably different due to the presence of free hemoglobin. To explain the experimental observations, Gautam et al. used a theoretical model with an optical force-mediated nonlocal nonlinearity. The present work on light self-guiding through scattered soft biological matter can introduce new photonic tools for noninvasive biomedical imaging and medical diagnosis.
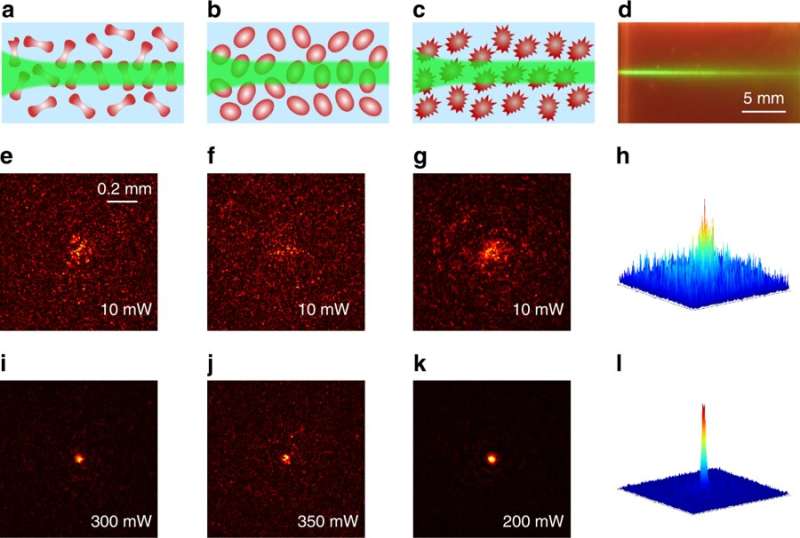
Human RBCs are disc-shaped malleable cells that possess a spatially uniform refractive index as they lack nuclei unlike most organelles, and show distinctive deformability for passage through veins and microcapillaries. The shape change can be prompted by modifying the osmolarity of the surrounding liquid buffer to use RBCs as tunable optofluidic microlenses. The optical properties of RBCs are important for in vitro and in vivo disease diagnostics in which the refractive index of the RBC is determined by hemoglobin (Hb)—the largest part of the erythrocyte dry content by weight. As a result, if the cell volume decreased due to varying osmotic conditions, the refractive index increased.
Pathophysiological conditions such as sickle cell anemia, malaria and sepsis are often closely related to the physical properties of RBCs, their shape and size. The fundamental features of varying refractive indices and cell shapes allow RBCs to react to changes in different osmotic environments making them ideal candidates to study scattering light. In the present work, Gautam et al. showed nonlinear self-trapping of light across a centimeter distance of propagation by scattering RBC suspensions. When they increased the power of the laser beam, they showed the beam dramatically self-focus within all three osmotic conditions – much like optical spatial solitons (nonlinear self-trapped wave packets). The optical forces that change with cell density and morphology can provide noninvasive tools to sort diverse cells, according to a specific stage of a given disease.
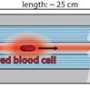
The scientists obtained blood samples from anonymous donors for the experiments. In the first set of experiments, they used a linearly polarized continuous wave (CW) laser beam with a wavelength of 532 nm. They focused the light into a 3 cm long glass cuvette filled with RBC suspensions in diverse osmotic conditions, as previously described. They monitored the linear and nonlinear outputs from the sample using a CCD camera and power detector, and measured the beam diameters using the Beamview program. The beam first diffracted normally at a low power of 10 mW and experienced strong scattering thereafter due to random distribution of non-spherically shaped RBCs.
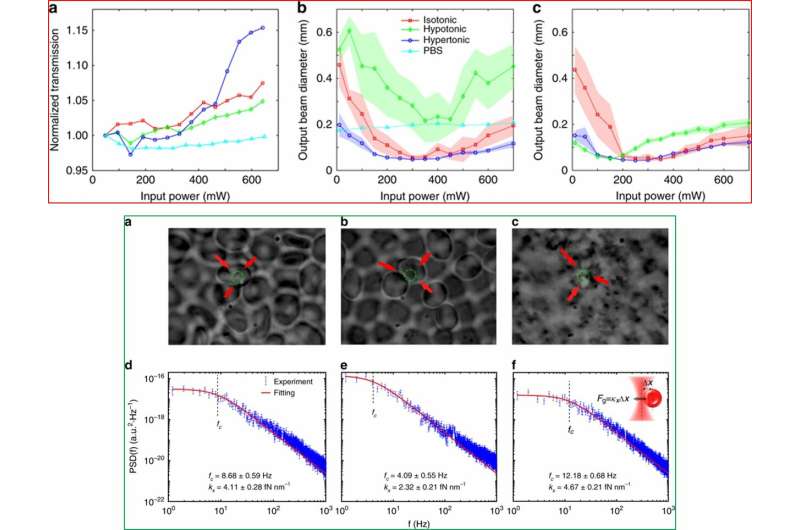
Gautam et al. then measured normalized laser transmission (output/input power) as a function of the input beam power. In hypotonic solutions, they noted the RBCs were in a "swollen" state where the effective refractive index of the cells decreased as the water-to-Hb ratio increased. In contrast, in the hypertonic solution, the scientists observed that RBCs shrunk, and their effective index increased due to reduced water-to-Hb ratio. In a third isotonic solution, the cells exhibited a "normal" state, in which the RBCs showed intermediate behavior. When the experiments were performed using the same blood samples two weeks later, the scientists observed notably different outcomes in which the nonlinear focus dramatically enhanced for the hypertonic solution.
In a second set of experiments, the scientists used a home-built optical tweezer system to measure the optical gradient force on RBCs. Gautam et al. collected the forward-scattering light from the trapped cells with a condenser lens and subsequently focused onto a position sensitive detector (PSD). They calculated the stiffness and gradient force in the three separate solutions. To simplify the measurements, Gautam et al. treated hypotonic and hypertonic RBCs as disk shaped objects. They used a CCD camera to record cell movements from the three different solutions along with a microscope with two objectives, where the setup was driven using a 960 nm laser beam. The results illustrated the movement of cells against Brownian motion under the action of optical forces based on the conditions of the cell (shape, size) and their beam trapping capacity. Gautam et al. estimated the trapping force using the Langevin equation and informed that the force followed a trend of hypertonic > isotonic > hypotonic conditions.
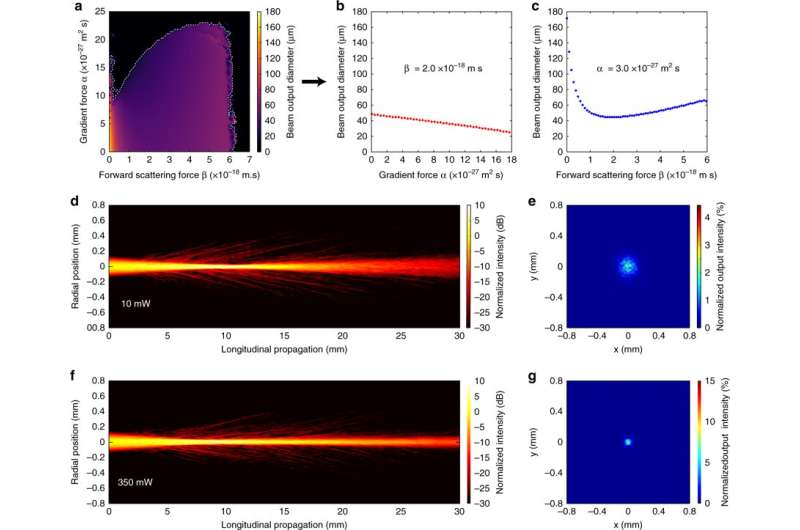
The scientists then developed a model to simulate nonlinear beam propagation in biological soft matter in order to understand the physics of optical force-mediated nonlinearity. They modelled time evolution of the particle concentration distribution using a diffusion-advection equation and considered the presence of a forward-scattering force to push the particles along the direction of beam propagation, alongside the optical gradient force. Gautam et al. calculated the change in beam size for the different gradient and scattering force parameters to simulate the nonlinear self-focusing effects under different buffer conditions. They recorded the changing size, volume and refractive indices of RBCs under diverse osmotic conditions that were accountable for the varying magnitude of optical forces that modified the optical nonlinearity. The simulated results were qualitatively consistent with the experimental observations.
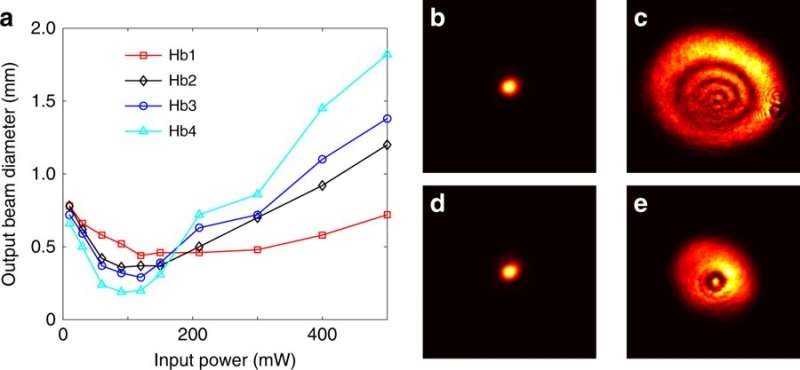
In this way, Gautam et al. studied nonlinear beam propagation in human RBCs suspended in three diverse buffer solutions. They found that RBCs exhibited a strong self-focusing nonlinearity that could be chemically controlled based on the buffer solution. They therefore propose tuning the optical nonlinearity via osmosis and increased osmotic pressure, outside the cells in fresh blood samples. When the samples aged, free hemoglobin from the lysed RBCs played an active role in the observed optical nonlinearity and enhanced the nonlinear response in hypotonic conditions.
Using direct video microscopy and optical tweezer measurements, the scientists showed that the beam trapping force was greatest for RBCs in the hypertonic conditions and weakest for hypotonic solutions. The scientists introduced a theoretical model to validate the observed experimental effects. The work will introduce a new perspective in the development of diagnostic tools as the results are very promising towards the development of laser treatment therapies for blood-related diseases.
More information: Rekha Gautam et al. Optical force-induced nonlinearity and self-guiding of light in human red blood cell suspensions, Light: Science & Applications (2019). DOI: 10.1038/s41377-019-0142-1
I. M. Vellekoop et al. Exploiting disorder for perfect focusing, Nature Photonics (2010). DOI: 10.1038/nphoton.2010.3
Roarke Horstmeyer et al. Guidestar-assisted wavefront-shaping methods for focusing light into biological tissue, Nature Photonics (2015). DOI: 10.1038/nphoton.2015.140
Roadmap on structured light. Journal of Optics. iopscience.iop.org/article/10. … 978/19/1/013001/meta
Journal information: Nature Photonics , Light: Science & Applications
© 2019 Science X Network