Shooting movies in atoms

Researchers of the Laboratory for Attosecond Physics at LMU and the Max Planck Institute for Quantum Optics have developed a microscope that tracks the motion of electrons.
The motion of electrons proceeds on timescales ranging from a few femtoseconds down to attoseconds. This makes them imperceptible to the human eye. Now, researchers of the Laboratory for Attosecond Physics (LAP) at LMU and the Max Planck Institute for Quantum Optics (MPQ) in Garching, Germany, have collaborated with the Joint Attosecond Laboratory (JASLab) in Ottawa, Canada, to develop a microscope that visualizes the motions of electrons. Using their laser-based method, the scientists are now able to film what happens inside atoms or molecules, when their electrons are excited by light.
"The key challenge in visualizing electrons is their speed," explains Dr. Matthias Kübel, a former member of Professor Matthias Kling's team at LMU. "In order to track their motion, we need to freeze it at very short intervals, as with a high-speed camera. We did this by using laser pulses that lasted less than five femtoseconds," he adds. The researchers applied femtosecond laser pulses to argon atoms, thus altering the behavior of their electrons. "It took less than 12 femtoseconds for the distribution of the electrons to change from the initial donut shape into a peanut shape," says Kübel. "While the electron motion is extremely fast, it is recurring, allowing us to monitor the reproducibility of our method."
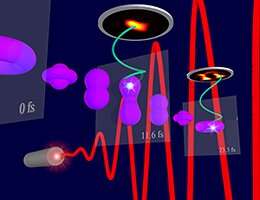
Using their microscope, the scientists showed how the electrons are distributed within an argon ion, and how their configuration changes with time. This was accomplished by directing two more laser pulses onto the excited argon ions generated by the first. In accordance with quantum mechanics, these laser pulses create a replica of the electron cloud inside the argon ions. This replica is imaged on a specialized electron detector. The individual images are then compiled by a computer to retrieve a movie of the electron motion. "This allows us to watch what happens within atoms or molecules immediately after they have interacted with light," says Kübel. (LAP/LMU)
More information: M. Kübel et al. Spatiotemporal imaging of valence electron motion, Nature Communications (2019). DOI: 10.1038/s41467-019-09036-w
Journal information: Nature Communications
Provided by Ludwig Maximilian University of Munich