Structure-ID technique could shift chemistry to warp speed

Work that previously could have taken chemists months can now be done in minutes.
Using a technique called microcrystal electron diffraction, or MicroED, scientists need just 30 minutes and a miniscule amount of sample to identify small molecules and determine their structures. Such easy access to highly detailed information could revolutionize the way chemists, forensic scientists, and those involved in drug discovery work, says Howard Hughes Medical Institute (HHMI) Investigator Tamir Gonen.
The technique uses a standard cryo-electron microscope and could become scientists' go-to procedure for identifying everything from the products of routine chemical reactions to unknown powders at crime scenes, he says. "Now chemists can actually take powders directly from a reaction, apply them to a sample grid, and get high-resolution molecular structures the same day."
Gonen's team published the structures of 11 small molecules, determined by MicroED, November 2, 2018 in the journal ACS Central Science. The paper builds upon small molecule structure work the scientists published earlier this year and demonstrates the scope of MicroED technology, Gonen says.
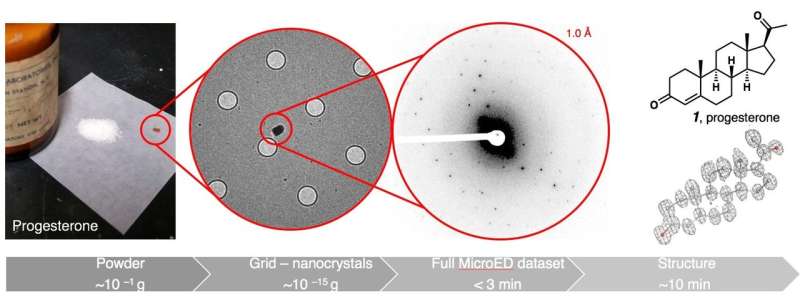
From powder to structure
Gonen's idea for the current project came over lunch with UCLA chemist Hosea Nelson. Gonen told him that, in the initial development of MicroED, his lab had determined the structure of a small organic molecule. Nelson, whose work as a chemist revolves around small molecules, "couldn't believe me when I told him it was pretty straightforward," Gonen says. So the two teamed up and decided to see how generally applicable MicroED was to chemistry and to alert the chemistry community of this technology.
Beginning with a jar of progesterone powder, they crushed a tiny amount and deposited it on a sample grid. Then, they cooled it to -196 degrees Celsius, transferred it to a cryo-electron microscope, and started collecting data. Less than 30 minutes passed from opening the jar to seeing progesterone's structure, Gonen says. His team tested eight more commercial powders and obtained similar results, even after mixing several of them together.
Knowing that the powders had probably all been crystallized during manufacturing, the team wanted to test compounds that were instead newly synthesized, and not crystallized by scientists. They mixed four of these compounds, separated them via a common purification technique, and analyzed them by MicroED. The technique produced structures for two of the four compounds, which had formed crystals spontaneously. Gonen believes the other two might have worked had the team tried to crystallize them first.
Gonen doesn't see X-ray crystallography or other structure-identification methods going away anytime soon. Some samples will be more amenable to one method over another, and the information each method supplies varies in useful ways, he says. But now, the natural ability of small molecules to form crystals can be harnessed by chemists in a way that wasn't before possible.
"This is a perfect example of what happens when two fields that normally don't talk to one another get together and cross-pollinate," Gonen says.
More information: Christopher G. Jones et al. The CryoEM Method MicroED as a Powerful Tool for Small Molecule Structure Determination, ACS Central Science (2018). DOI: 10.1021/acscentsci.8b00760
Journal information: ACS Central Science
Provided by Howard Hughes Medical Institute