Chemists report a detailed description of azopyrazolonic dyes
RUDN chemists have identified the structure of azopyrazolone dyes. The new development will make their synthesis more eco-friendly. The work of the scientists was published in Reviews in Inorganic Chemistry.
Pyrazolones are chemical compounds that contain pyrazolone cycles. They are used in the pharmaceutical industry as basic substances for medicinal drugs, specifically anaesthetics. By adding nitrogen atoms, so-called azopyrazolonic dyes can be isolated. These are used as synthetic dyes for textiles. They have intensive coloration and are resistant to mechanical and light treatment. However, the production of azopyrazolone dyes with required properties is not 100 percent successful. Sometimes, the obtained results are not fit for further use. The reasons for this have been unknown until recently. To understand them, RUDN chemists carried out detailed analysis of the chemical and geometrical structure of azopyrazolonic dye molecules.
"Some new dyes are applicable in textile manufacture, and some are not. The question is why. To synthesize targeted chemical compounds with pre-set qualities, we have to understand the reason, to know the spatial orientation of their molecules and the groups and fragments with which these molecules are attached to the surface of textiles," explains Olga Kovalchukova, Professor of the Department of General Chemistry, RUDN.
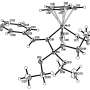
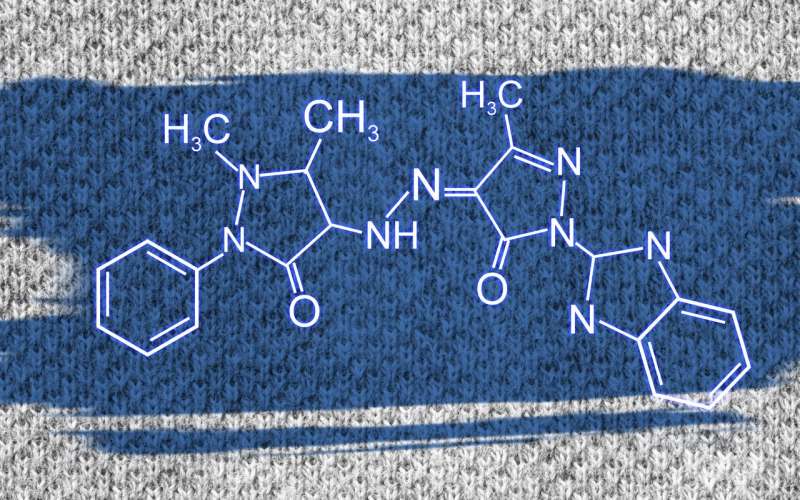
RUDN chemists were the first to summarize all scientific publications covering this topic and to describe the structure of azopyrazolone dyes based on their own experiments. Previously, chemists supposed that azopyrazolone dyes existed in two forms—azo-form and hydrazo-form. Due to a lack of data, the forms were considered mutually transitional, i.e. turning into each other under certain physical conditions. RUDN chemists now provide a better explanation.
"Having summarized all publications regarding this topic, we found that the substances had one and the same form, that is hydrazo-form. Differences in behavior are explained by spatial orientation of molecules which are quite long," said Kovalchukova.
The understanding of the structure of azopyrazolonic dyes will help optimize synthesis technologies. Previously, the manufacture of the dyes took up to 20 to 30 chemical processes each, contributing to air and waste water pollution. The work by the RUDN chemists will make synthesis more targeted and reduce the number of reactions to just one.
"We've got better understanding of the structure of azopyrazolonic dyes, and this will help us envisage the properties of new dyes. Previously, these substances were synthesized in accordance with outdated concepts and sometimes haphazardly, but starting from now, we'll be able to foresee the structure of the molecules and their properties on a higher level. When you know in what direction to move, you don't need 20 or 30 synthetic processes—just one is enough," concluded Kovalchukova.
More information: Olga V. Kovalchukova et al. Tautomeric transformations and electronic structures of azopyrazolone dyes and their metal complexes, Reviews in Inorganic Chemistry (2018). DOI: 10.1515/revic-2018-0007
Provided by RUDN University