Stabilizing silver films for high-efficiency fuel cells

Solid oxide fuel cells (SOFCs) offer a stable and efficient way to generate clean electrochemical power, but are impractical for use in portable devices because of their high operating temperatures. A new design and production strategy developed by Florencia Edith Wiria of the A*STAR Singapore Institute of Manufacturing Technology and Pei-Chen Su at Nanyang Technological University could help propel SOFCs into mainstream use1.
Micro-scale SOFCs could transform consumer electronics, delivering more power than existing batteries in an environmentally-friendly way. Silver is an appealing and affordable alternative to the costly platinum cathodes employed in current micro-SOFC designs. But silver electrodes melt, so they cannot retain the fine porous structure required for an efficient electrochemical reaction at the 500-1,000 degrees Celsius temperature range at which these devices generally operate. Wiria and Su therefore sought to develop a heat-resistant version of this system.
"This would enable the extension of SOFCs from conventional stationary power sources to portable applications," says Wiria.
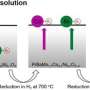
Wiria and Su used a strategy called 'wet chemical infiltration', in which they coated thin films of silver with a layer of samarium-doped ceria (SDC). Critically, their approach made use of a 3-D printer, conferring exquisite control over the electrode design. "We wanted to leverage the capability to print fine, complex structures to realize power sources with various shapes," says Wiria. The resulting silver films retained the desired nanoscale structure, but were also protected under a crystalline layer of SDC.
While conventional silver films quickly melted into a formless aggregate as temperatures climbed past 300–400 degrees Celsius, the SDC-infiltrated films remained largely unchanged even at 500 degrees Celsius. This enhanced cathode integrity translated into robust fuel cell performance after more than a day of continuous operation, with performance that even surpassed platinum electrodes. "We significantly improved the thermal stability of solid-oxide fuel cells with nanoporous silver cathodes from current degradation of 73.6 per cent to only 7.9 per cent," says Wiria.
Microscopic analysis confirmed that the electrode microstructure remained largely intact even after this test, and the infiltrated cathodes exhibited only modest additional degeneration after 60 hours of operation.
This work promises to greatly extend the utility of micro-scale SOFCs, and Wiria and Su are looking into further modifications that could confer even greater stability and flexibility in their cathode designs. "We are currently trying to use a 'core-shell' method to fully encapsulate our silver nanoparticles," explains Wiria, "and looking into other 3-D printing methods to produce SDC-infiltrated SOFCs."
More information: Tsung-Han Lee et al. A high-performance SDC-infiltrated nanoporous silver cathode with superior thermal stability for low temperature solid oxide fuel cells, Journal of Materials Chemistry A (2018). DOI: 10.1039/C8TA01104D
Journal information: Journal of Materials Chemistry A