How sleeping mammary stem cells are awakened in puberty
Walter and Eliza Hall Institute researchers have discovered how the growth of milk-producing mammary glands is triggered during puberty.Sleeping stem cells in the mammary gland are awoken by a protein dubbed FoxP1, according to the research that was published today in the journal Developmental Cell.
The research expands our knowledge of how the mammary gland—a component of the human breast—develops from stem cells, underpinning a better understanding of how defects in this process lead to breast cancer. The research was led by Dr. Nai Yang Fu, Professor Jane Visvader and Professor Geoff Lindeman who is also a medical oncologist at the Royal Melbourne Hospital and the Peter MacCallum Cancer Centre, in collaboration with Professor Gordon Smyth and his bioinformatics team.
Waking up stem cells
Stem cells in the mammary gland exist in a largely dormant or 'sleeping' state throughout life. In puberty, these stem cells need to be 'woken up' to drive the rapid expansion of the mammary gland, said Professor Visvader.
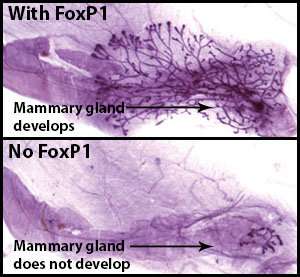
"The mammary stem cells are ready for a signal to start dividing," she said. "We discovered that a gene called FoxP1 is an essential part of this signal in puberty and the adult."
FoxP1 switches off the production of other proteins within cells—by repressing their genes.
"We discovered that FoxP1 switches off the production of one of the key proteins that keep mammary stem cells asleep. As the level of this protein drops, the stem cells wake up and begin to divide, driving mammary gland growth," Dr. Fu said.
The importance of team work
The project relied on collaboration between scientists with diverse skills, said Professor Visvader.
"This project brought together expertise in cell biology, developmental biology, bioinformatics and imaging to solve the question of how mammary stem cells are awoken in puberty and adult breast tissue.
"We're still looking for the precise connections linking female hormones and FoxP1, but we are one step closer to understanding the detailed process of breast development. This is also helping us to connect faulty cells that contribute to breast development with the development of breast cancer," she said.
More information: Developmental Cell (2018). DOI: 10.1016/j.devcel.2018.10.001
Journal information: Developmental Cell
Provided by Walter and Eliza Hall Institute