Regulating gene transcription using light
Researchers led by Mustafa Khammash have developed a new method that uses blue light to control the transcription of DNA into RNA in single cells. The technology could also be used in tissue engineering and stem cell research.
Transcription is a fundamental biological process in which a gene is copied to an RNA molecule. This involves proteins, including transcription factors and a molecular machine called RNA-polymerase.
For the transcription process to begin, the transcription factors must first attach to a specific part of the DNA. This triggers various processes that lead to the docking of the RNA polymerase to the DNA. This protein complex then untwists the double helix in order to read the sequence of the gene and copy it into an RNA molecule. The so-called messenger RNA is a transportable copy of the gene that functions as a template for protein synthesis.
Despite being strongly regulated by many molecular players, transcriptional output often exhibits high levels of variability between cells. This variability results partly from the fact that transcription is based on the random encounter of molecules, such as transcription factors and a specific DNA sequence. Transcription of a specific gene can therefore proceed differently in each cell; it starts more quickly in some and not at all in others. Achieving a better understanding of the sources of this variability and gaining greater control of the transcription process is an important research objective.
The research group led by Mustafa Khammash, Professor of Control Theory and Systems Biology at the Department of Biosystems and Engineering in Basel, has now found a new way to control when transcription is initiated, and regulate the amount of RNA molecules formed using an optogenetic platform, which the ETH researchers recently presented in the journal Molecular Cell. This enables variability between cells to be reduced and allows new insights into transcriptional dynamics.
Individual feedback, individual regulation
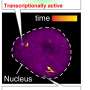
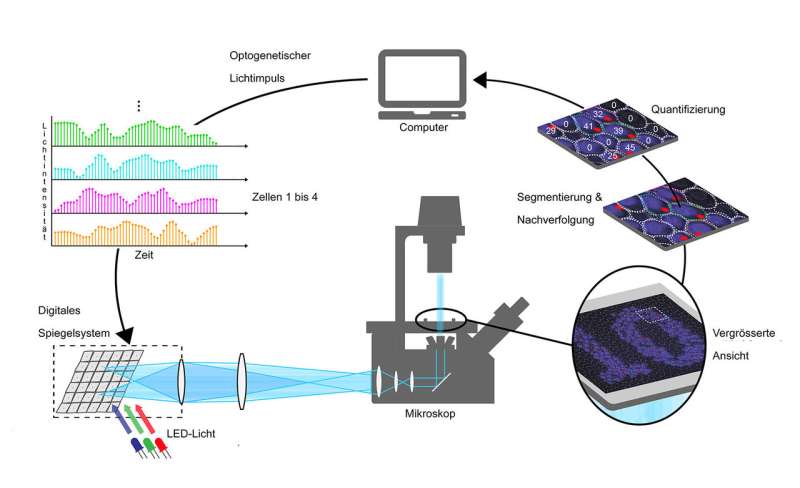
The platform allows the researchers to illuminate single yeast cells with blue light for varying durations and intensities. Through the introduction of foreign genes, these cells have been genetically engineered to respond to light of this colour by activating a transcription factor and thereby promoting the transcription of a specific gene. The transcripts of this gene, the messenger RNAs, are tagged with fluorescent proteins, which results in bright fluorescent spots when transcription is actively occurring.
These spots can be imaged and evaluated using fluorescence microscopy. The researchers linked this system to a computer, which counts how many RNA molecules are transcribed at a given time and decides on the amount of light that each cell should receive next, in order to regulate their transcription as desired or as specified.
New insights on transcriptional dynamics
"Thanks to this set-up, we were able to show that transcriptional activation and deactivation happens very quickly," says ETH Professor Khammash. His team further established that transcription occurs in bursts, whose duration and timing are modulated by transcription factor activity. Thanks to their feedback mechanism, the researchers were also able to reduce the differences in transcriptional output between different cells.
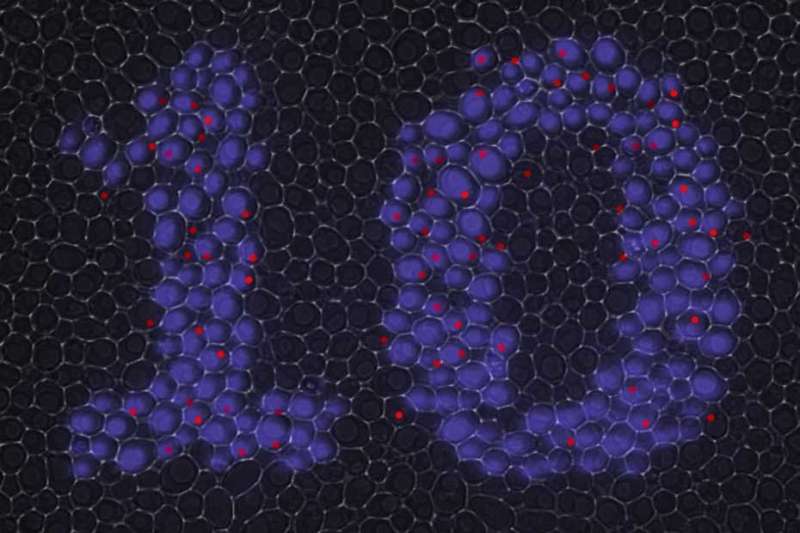
As a test (and to celebrate the 10th birthday of the Department for Biosystems last year), the researchers projected the number 10 onto yeast cells using blue light. In response to the light, transcription was initiated in the illuminated cells which resulted in the formation of bright fluorescent spots at the desired positions.
Dynamic signals reduce variability
The control method described has a limitation: it can only be used under the microscope, but not in test tubes or in bioreactors, where it would be needed to control biotechnological processes.
To overcome this limitation, the researchers turned to dynamic signalling. In contrast to conventional methods of controlling gene activity, the use of light allows cells to be controlled using complex, dynamic signals. By comparing different types of signals, the researchers observed in a study published in Nature Communications that individual cells react to pulsatile signals with higher accuracy than to constant signals. This now makes it possible to better control large populations of cells in a simple way. Furthermore, the results hint at a possible strategy by which variability may be regulated in natural cell populations.
Khammash is delighted that the projects were a success. He and his colleagues initially began this research out of pure curiosity, without any specific applications in mind. "We wanted, above all, to find out if we could control the random element of the transcription process," he said. "It was a real technical challenge."
He now thinks that his platform could be a gold mine for follow-up studies. He says that there could be many different applications, particularly in research, where controlling transcription is important. As an example, prototypes of genetic networks could be developed with increased speed, as cell-to-cell communication could be controlled externally and would no longer be reliant on signaling molecules. "The platform could also become an important tool for tissue engineering and stem cell research." The researchers will take a closer look at these applications of their platform over the next few years, and have already secured funding from the EU FET open program to do so.
More information: Marc Rullan et al. An Optogenetic Platform for Real-Time, Single-Cell Interrogation of Stochastic Transcriptional Regulation, Molecular Cell (2018). DOI: 10.1016/j.molcel.2018.04.012
Dirk Benzinger et al. Pulsatile inputs achieve tunable attenuation of gene expression variability and graded multi-gene regulation, Nature Communications (2018). DOI: 10.1038/s41467-018-05882-2
Journal information: Nature Communications , Molecular Cell
Provided by ETH Zurich