Brain-eating amoebae halted by silver nanoparticles
Halloween is just around the corner, and some people will celebrate by watching scary movies about brain-eating zombies. But even more frightening are real-life parasites that feed on the human brain, and they can be harder to kill than their horror-movie counterparts. Now, researchers have developed silver nanoparticles coated with anti-seizure drugs that can kill brain-eating amoebae while sparing human cells. The researchers report their results in ACS Chemical Neuroscience.
Although infections with brain-eating amoebae (Naegleria fowleri) are rare, they are almost always deadly. Most cases result from inhaling warm, dirty water in ponds, hot springs or unchlorinated swimming pools. Another species, Acanthamoeba castellanii, can cause blindness by entering the eyes through dirty contact lenses. Common treatments include antimicrobial drugs, but they often cause severe side effects because of the high doses required for them to enter the brain. Ayaz Anwar and colleagues wondered if three anti-seizure drugs—diazepam, phenobarbitone and phenytoin—could kill amoebae, alone or in combination with silver nanoparticles. The drugs are already approved by the U.S. Food and Drug Administration and are known to cross the blood-brain barrier. The researchers reasoned that they might be more effective when attached to silver nanoparticles, which can improve the delivery of some drugs and also have their own antimicrobial effects.
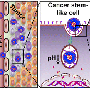
The team chemically attached the drugs to silver nanoparticles and examined their ability to kill amoebae. They found that each of the three drugs alone could kill N. fowleri and A. castellanii, but they worked much better when bound to silver nanoparticles. The drug-nanoparticle combos protected human cells from the microbes, increasing their survival rate compared with untreated infected human cells. The researchers propose that these repurposed drugs, aided by the nanoparticles, might kill amoebae by binding to protein receptors or ion channels on the single-celled organism's membrane.
More information: Ayaz Anwar et al. Clinically-approved drugs against CNS diseases as potential therapeutic agents to target brain-eating amoebae, ACS Chemical Neuroscience (2018). DOI: 10.1021/acschemneuro.8b00484
Abstract
Central nervous system (CNS) infections caused by free-living amoebae such as Acanthamoeba species, Naegleria fowleri etc. are rare but fatal. A major challenge in the treatment against the infections caused by these amoebae is the discovery of novel compounds that can effectively cross the blood-brain barrier to penetrate CNS. It is logical to test clinically-approved drugs against CNS diseases for their potential antiamoebic effects since they are known for effective blood-brain barrier penetration and effect eukaryotic cell targets. The antiamoebic effects of clinically available drugs for seizures targeting gamma-amino butyric acid (GABA) receptor and ion channels were tested against Acanthamoeba castellanii (A. castellanii) belonging to the T4 genotype and Naegleria fowleri (N. fowleri). Three such drugs namely; Diazepam (Valium), Phenobarbitone (Luminal), Phenytoin (Dilantin) and their silver nanoparticles (AgNPs) were evaluated against both trophozoites and cysts stage. Drugs alone and drugs conjugated silver nanoparticles were tested for amoebicidal, cysticidal and host-cells cytotoxicity assays. In vitro amoebicidal assay showed potent amoebicidal effects for Diazepam, Phenobarbitone, and Phenytoin-conjugated AgNPs as compared to drugs alone against A. castellanii and N. fowleri. Nanoparticles were synthesized by sodium borohydride reduction of silver nitrate with drugs as capping agents. Drugs conjugated nanoconjugates were characterized by ultraviolet-visible (UV-vis), and Fourier transform infrared (FT-IR) spectroscopies, and atomic force microscopy (AFM). Furthermore, both drugs and drugs conjugated AgNPs showed compelling cysticidal effects. Drugs conjugations with silver nanoparticles enhanced their antiacanthamoebic activity. Interestingly, amoeba-mediated host cells cytotoxicity was also significantly reduced by drugs alone as well as their nanoconjugates. Since, these drugs are being used to target CNS diseases, their evaluation against brain-eating amoebae seems feasible due to advantages such as; permeability of the blood-brain barrier; established pharmacokinetics and dynamics; FDA approval etc. Given the limited availability of effective drugs against A. castellanii, the clinically available drugs tested here present potential for further in vivo studies.
Journal information: ACS Chemical Neuroscience
Provided by American Chemical Society