Sex in plants requires thrust

In a paper to be published in the September 2018 issue of TECHNOLOGY, the thrust produced by the microscopic organ delivering sperm cells in plants has now been measured using microfluidic technology. A team of nano and microsystems engineers and plant biologists at the University of Montreal (now known as Université de Montréal), McGill University and Concordia University devised a microchip that enabled the researchers to measure how much force the sperm delivery tools used by plants exert when negotiating the female flower tissues to accomplish fertilization.
The fabrication of the lab-on-a-chip used to measure the growth force of the pollen tubes was highly complex, involving the assembly of multiple layers of precisely designed elastomer material with a precision in the micrometer range. Reproduction in plants is crucial for human survival, since most of the calories we consume depend on it. Successful plant fertilization is at the basis of the formation of seeds such as rice, corn and wheat, as well as fruits and numerous vegetables. Failure to fertilize can therefore have dramatic economic and humanitarian consequences.
To mimic the biological maze of the flower tissue, the pollen tubes were strategically guided into the microdevice and exposed to miniature force balances. When the tubes interacted with the gauge, they deflected the device and the stalling force provided information on the amount of thrust the tiny cells were able to exert. The magnitude of this force suggests that the cellular pressure driving the invasion process corresponds to that of a typical car tire. Curiously, the pollen tubes changed the frequency or their oscillatory growth pattern, suggesting that the cells "feel" and respond to the physical resistance of the environment.

Sperm cell delivery in plants has many similarities with the analogous process in humans and animals. The egg cell in plants is deeply embedded in the female tissues, and hence, the delivery of the sperm cells by the male partner, the pollen grain, requires the formation of a protuberance that invades the female tissues. Contrary to the human counterpart, each plant male only delivers two sperm cells, and the process ends with its death. "This is the reason that flowers form so many pollen grains" says senior author of the study, Anja Geitmann. The biological purpose of these microscopic spheres is the delivery of sperm cells from one flower to the next.
Unlike animal sperm cells, plant sperm cells are not motile, and therefore they have to be delivered directly to the egg cell for fertilization to occur. The growth of the delivery organ is therefore crucial. The female flower tissues represent a maze of formidable complexity through which the pollen tubes have to manoeuvre. Essential for targeting, the pollen tube is able to follow the guidance cues emitted by the female partner. "We have long suspected that the process requires not only a sophisticated navigation system, but also substantial forces," explains Geitmann, "but just how much force this single cell can produce was unknown."
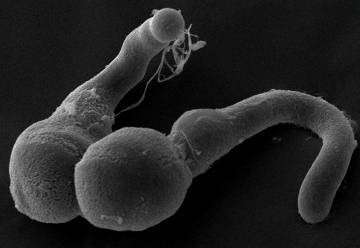
Concordia University Professor Muthukumaran Packirisamy adds, "From a mechanical point of view, the process of pollen tube elongation is similar to that of a balloon catheter used for angioplasty—forces are generated based on the principle of a hydroskeleton, or fluid under pressure. We designed a microscopic cantilever-based gauge against which the pollen tubes had to forcefully push in order to continue their elongation."
Microsystems technology was required for this experiment due to the tiny size of pollen tubes and the consequently minute amount of force they exert to accomplish penetration. In big flowers, pollen tubes can become many centimeters long, but independently of flower size, the tubes are only between five and 20 micrometres wide. By way of comparison, a human hair is typically 100 micrometres thick.
The study was designed not only to quantitatively determine the invasive force of the pollen tube, but also to closely monitor its behavior upon the encounter with the physical object. The researchers observed that the characteristic oscillatory growth pattern of the cell was markedly altered by contact with the physical obstacle leading them to suspect that the tube perceives and is able to adapt its growth force depending on the mechanical impedance it encounters. Such tunable and sophisticated behavior by a single cell is fascinating and illustrates the complexity of the reproductive process, even in plants.
The team from McGill University is working now to further characterize the invasive behavior of the pollen tube to understand how the cellular growth machinery steers the growth direction toward its target, and how the cell interacts with the female tissues while the team at Concordia University continues the development of lab-on-a-chip for studying varying cellular behavior and properties.
More information: Mahmood Ghanbari et al, Measuring the growth force of invasive plant cells using Flexure integrated Lab-on-a-Chip (FiLoC), TECHNOLOGY (2018). DOI: 10.1142/S2339547818500061
Provided by World Scientific Publishing