The origin of off-taste in onions
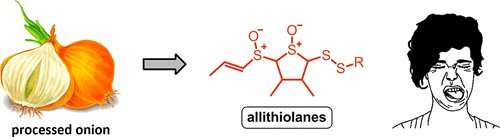
Chopping onions is usually associated with watery and stinging eyes. But after the onions are diced and the tears are dried, the vegetable pieces can sometimes develop an unpleasant bitter taste. Now, one group reports in ACS' Journal of Agricultural and Food Chemistry that they have identified previously unknown compounds causing this off-taste.
Thanks to onions' characteristic taste and strong smell, they are one of the most popular vegetables around the world. Onions are known for their versatility—people frequently eat them raw or add them to numerous dishes as they cook. But as onions are chopped and processed for cooking, a bitter taste that could affect the taste of the resulting meal can sometimes develop. Very little is known about why this particular phenomenon occurs. Previous research has shown that onions release a compound called lachrymatory factor upon being cut that causes eyes to water and sting. In addition, scientists have also concluded that several sulfur-based compounds are also formed when the onion is chopped, but none of these caused a bitter taste. So Roman Kubec and colleagues wanted to identify the bitter onion compounds.
The team processed onions with a kitchen juicer. Freshly obtained juice was not bitter, but after 30 minutes, it developed a strong bitter taste. The group performed sensory-guided high-performance liquid chromatography to identify the compounds that formed over time in the onion juice. The researchers discovered nine groups of new sulfur compounds in the onion juice, which they dubbed allithiolanes. These compounds form spontaneously when the onion is damaged. The allithiolanes were not limited to onions; further testing showed they were also present in leeks, and one of the groups was found in garlic.
More information: Roman Kubec et al. Allithiolanes: Nine Groups of a Newly Discovered Family of Sulfur Compounds Responsible for the Bitter Off-Taste of Processed Onion, Journal of Agricultural and Food Chemistry (2018). DOI: 10.1021/acs.jafc.8b03118
Abstract
The compounds responsible for the bitter off-taste of processed onion (Allium cepa) were studied. Using a series of sensory-guided HPLC fractionations, the existence of nine groups of hitherto unknown sulfur compounds has been revealed. On the basis of spectroscopic data (MS, NMR, and IR), it was found that these compounds, trivially named allithiolanes A–I, are members of a large family of structurally closely related derivatives of 3,4-dimethylthiolane S-oxide, with the general formulas of CxHyO2S4, CxHyO3S5, and CxHyO4S6 (x = 10–18, y = 18–30). The presence of multiple stereoisomers was observed for each group of allithiolanes. Allithiolanes possess an unpleasantly bitter taste with detection thresholds in the range of 15–30 ppm. Formation pathways of these newly discovered sulfur compounds were proposed.
Journal information: Journal of Agricultural and Food Chemistry
Provided by American Chemical Society