The nitty-gritty behind how onions make you cry
Adding onions to a recipe can make a meal taste rich and savory, but cutting up the onion can be brutal. Onions release a compound called lachrymatory factor (LF), which makes the eyes sting and water. Scientists know that a certain enzyme causes this irritating compound to form but precisely how it helps LF form in the onion remained an open question. Now, one group reports in ACS Chemical Biology that they have the answer.
According to the National Onion Association, the average American consumes 20 pounds of onions each year. When an onion is cut, it has a natural defense mechanism that springs into action, producing LF. This kind of compound is rare—only four known natural types exist. An enzyme in the onion known as lachrymatory factor synthase (LFS) spurs production of LF in the onion. "Tearless" onions, sold exclusively in Japan for a hefty price, don't make LFS so they also don't produce the irritant LF. But scientists have been at a loss to explain exactly how LFS helps LF form. That's because it is extremely reactive, and LF evaporates or breaks down easily. Marcin Golczak and colleagues wanted to take a different approach to solve this mystery once and for all.
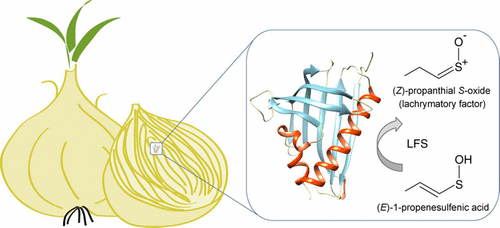
The team determined the crystal structure of LFS and analyzed it. With the crystal structure, they could finally see the architecture of the enzyme as a whole and its active site as it bound to another compound. By combining this information with known information about similar proteins, the group developed a detailed chemical mechanism that could explain the precise steps involved in LF synthesis—and hence, why people wind up crying when they chop an onion.
More information: Josie A. Silvaroli et al. Enzyme That Makes You Cry–Crystal Structure of Lachrymatory Factor Synthase from Allium cepa, ACS Chemical Biology (2017). DOI: 10.1021/acschembio.7b00336
Abstract
The biochemical pathway that gives onions their savor is part of the chemical warfare against microbes and animals. This defense mechanism involves formation of a volatile lachrymatory factor (LF) ((Z)-propanethial S-oxide) that causes familiar eye irritation associated with onion chopping. LF is produced in a reaction catalyzed by lachrymatory factor synthase (LFS). The principles by which LFS facilitates conversion of a sulfenic acid substrate into LF have been difficult to experimentally examine owing to the inherent substrate reactivity and lability of LF. To shed light on the mechanism of LF production in the onion, we solved crystal structures of LFS in an apo-form and in complex with a substrate analogue, crotyl alcohol. The enzyme closely resembles the helix-grip fold characteristic for plant representatives of the START (star-related lipid transfer) domain-containing protein superfamily. By comparing the structures of LFS to that of the abscisic acid receptor, PYL10, a representative of the START protein superfamily, we elucidated structural adaptations underlying the catalytic activity of LFS. We also delineated the architecture of the active site, and based on the orientation of the ligand, we propose a mechanism of catalysis that involves sequential proton transfer accompanied by formation of a carbanion intermediate. These findings reconcile chemical and biochemical information regarding thioaldehyde S-oxide formation and close a long-lasting gap in understanding of the mechanism responsible for LF production in the onion.
Journal information: ACS Chemical Biology
Provided by American Chemical Society