Efficient synthesis of multi-substituted anilines by domino rearrangement
Anilines have been widely used in medicine, in particular in acetaminophen pain killers. They are also used in organic materials, such as liquid crystals and organic light-emitting diodes. The ability to efficiently synthesize a new class of aniline derivatives will enable further developments in medical and materials science.
In a recent study, Yasuhiro Ishida, Itaru Nakamura, and Masahiro Terada at Tohoku University have reported that anilines, in which more than two substituents were placed sequentially, were synthesized from readily accessible substrates (ortho-alkylated N-methoxyanilines) in a selective manner.
Until now, preparation of multiply substituted anilines by conventional synthetic methods has been inefficient due to low selectivity.
Researchers found the reaction in the current study was effectively promoted by cationic copper catalysts under mild reaction conditions.
Substrates which have a substituent at the para position were efficiently converted to the desired anilines in which the four substituent are sequentially arranged, in good yields. Since conventional methods for synthesizing multi-substituted anilines have required multiple steps and often suffered from low efficiency, the new method offers vast improvement.
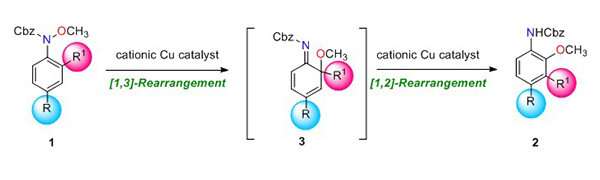
Mechanistic studies suggest that the unique transformation is regarded as "domino rearrangement," in which the first [1,3] rearrangement of the methoxy group induces the second [1,2] rearrangement from the ortho position to the meta position.
It is expected that the products obtained by the new method will contribute to the development of medicine and organic materials. Further development of the catalytic domino rearrangement reaction will lead to fewer processes and selective synthesis of a wide variety of multi-substituted aromatic compounds.
More information: Yasuhiro Ishida et al. Copper-Catalyzed Domino [1,3]/[1,2] Rearrangement for the Efficient Synthesis of Multisubstituted ortho-Anisidines, Journal of the American Chemical Society (2018). DOI: 10.1021/jacs.8b03669
Journal information: Journal of the American Chemical Society
Provided by Tohoku University