New technique can track drug and gene delivery to cells
With targeted drug and gene therapies, finding the target cells is only half the battle. Once these agents reach a cell's surface, they still have to get inside and do their job.
University of Illinois researchers say they now know how to track and map drug and gene delivery vehicles to evaluate which are most effective at infiltrating cells and getting to their targets, insight that could guide development of new pharmaceutical agents. The researchers described their tracking system and their findings on the most effective delivery vehicles in the journal Nature Communications.
Gene therapies have shown promise in cell culture and animal studies but have been less effective in clinical trials, said study leader Andrew Smith, a professor of bioengineering at the U. of I. This class of pharmaceuticals, called biologics, are different from traditional drugs in that they need to be attached to specialized delivery agents, such as nanoparticles or proteins, to reach their intended cellular targets.
Their lack of efficacy stems from their difficulty in reaching targets within cells, and the obstacles hampering them are poorly understood, Smith said.
"We have these really great models that tell us how classical drugs work, but there's no model that works for these new biologics that have to have some additional mechanism to deliver to cells. This has been a key missing part of pharmaceutical medicine," Smith said. "If we don't understand the mechanisms of the problem, we can't solve it. Now we can pinpoint why that happens and figure out how to overcome the key bottlenecks, which has never been possible."
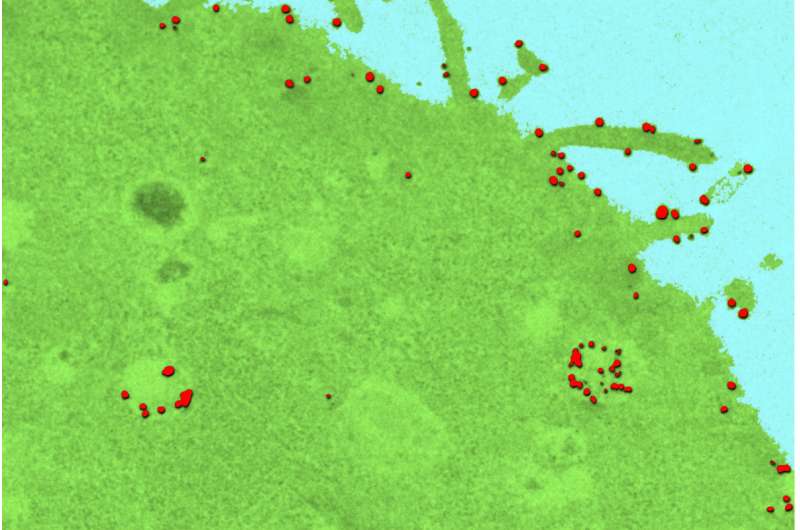
Smith's group combined two imaging tools to track and measure how different delivery agents entered human and mouse cancer cells. The researchers used single-molecule imaging, which allows them to observe individual molecules over time, and quantum dots, which act like tiny beacons inside cells. This allowed the researchers to see, count and track all of the delivery agents that entered the cell.
They found that molecules that are very charged are adept at getting inside cells but fail to reach their intended targets once inside.
"DNA is really charged, which is unfortunate, because that's one of the major things people are trying to deliver to cells," Smith said. "We see that it sticks to everything and doesn't get to its target."
However, molecules that are completely balanced in charge – a class called zwitterions – diffuse throughout the cell and can gain access to all of its machinery.
"It gives us some design rules for better ways to deliver genes and proteins to cells. We should avoid certain features and prioritize others – especially high-stability and zwitterionic surfaces," Smith said. "Anyone working in this area now can use this tracking technology to analyze new types of delivery agents. We only looked at a handful of cell types in our study, focusing on cancer, but these tools can evaluate any new agent that's developed for any cell type. You just need a high-quality fluorescence optical microscope, and all major research institutes have them."
The study used "empty" delivery agents – those without therapeutic cargo attached – so the next step is to use the system to track the agents actually delivering drugs or genes and observe the mechanisms along each step of the drug delivery pathway, Smith said.
"We are getting closer to solving the problem of cell delivery. This has been something that's limited medicine and gene therapy for a century," said Smith, who also is a professor in the Carle Illinois College of Medicine. "I think that the ability to track and analyze delivery in cells will be a huge boost to the pharmaceutical industry as it develops these types of therapeutics."
More information: Mohammad U. Zahid et al. Single quantum dot tracking reveals the impact of nanoparticle surface on intracellular state, Nature Communications (2018). DOI: 10.1038/s41467-018-04185-w
Journal information: Nature Communications
Provided by University of Illinois at Urbana-Champaign




















