Scientists find a new way to make novel materials by 'un-squeezing'
Some materials can morph into multiple crystal structures with very different properties. For instance, squeezing a soft form of carbon produces diamond, a harder and more brilliant form of carbon. The Kurt Vonnegut novel "Cat's Cradle" featured ice-nine, a fictional form of water with a much higher melting point than regular ice that threatened to irreversibly freeze all the water on Earth.
These materials are called polymorphs, and they're commonly made by applying pressure, or squeezing. Scientists looking for new materials would also like to do the opposite – apply "negative pressure" to stretch a material's crystal structure into a new configuration. In the past, this approach seemed to require negative pressures that are difficult if not impossible to achieve in the lab, plus it risked pulling the material apart.
Now researchers at the Department of Energy's National Renewable Energy Laboratory (NREL) have found a way to create the equivalent of negative pressure by mixing two materials together under just the right conditions to make an alloy with an airier and entirely different crystal structure and unique properties.
X-ray measurements at DOE's SLAC National Accelerator Laboratory confirmed that they have succeeded. Their new alloy has more space between its atoms then either of its parent materials, as if it had been stretched out. Unlike its parents, the new material is piezoelectric – able to generate an electric charge in response to applied mechanical stress. This may be important for use in sensors and actuators.
Published in Science Advances, the paper reporting their findings contains both the theoretical understanding and experimental proof of concept that show how such novel low-density materials can be made.
"Creating the negative pressure conditions in alloys required counterintuitive thinking, and maybe that's why this has not been pointed out before," said Andriy Zakutayev, an NREL scientist and a lead author of the new paper.

"It's possible to apply positive pressure to the material by squeezing it, but it's really difficult to un-squeeze the material," he said. "Think about it this way: you can pack a snowball out of snow, but you can't turn a dense snowball back into fluffy snow."
Pioneering Work on Mismatched Materials
The experiment builds on pioneering work led by NREL on mixing compounds with atomic structures that didn't match.
As surely as water flows downhill, a chemical reaction will take the path of least resistance, proceeding in a way that consumes the least energy. Mixing two materials with high-density structures takes a lot of energy; but if you could nudge the reaction in the direction of creating a low-density end product whose atoms were farther apart, it would require a lot less energy, the researchers theorized. It would be the equivalent of making a material under negative pressure.
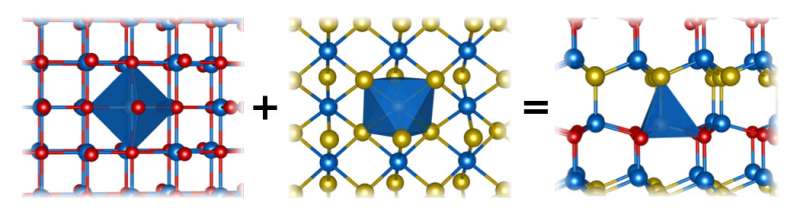
They tested this hypothesis by mixing high-density forms of manganese selenide and manganese telluride that have different crystal structures – one resembling rock salt, the other the mineral nickeline. To combine the two, they used a technique called sputtering in which fine sprays of atoms were freed from the surfaces of both starting materials and deposited as a thin film on a hot surface, where the new alloy crystallized and grew, said SLAC associate staff scientist Laura Schelhas. She conducted X-ray measurements of the new alloy at the lab's Stanford Synchrotron Radiation Lightsource (SSRL) with staff scientist Kevin Stone. The alloy, it turned out, had the crystal structure of yet another mineral, wurtzite.
"You can use this alloying approach to make different, never-before-seen materials with interesting properties," Schelhas said. "For instance, this one becomes piezoelectric: When you apply a voltage across the material it actually stretches and gets bigger. And the opposite also happens: If you push on it, it produces an electric charge. These piezoelectric materials are used in a lot of places – including some airbags, where the pressure from getting hit in a collision creates a little bit of electric charge that sets the airbag off – and the material created here was entirely new."
Aaron Holder, an NREL researcher and assistant professor at the University of Colorado Boulder, said, "The properties of this new alloy could lead to its use as a contact layer for solar cells, or for future magnetic memory, thin film transistors, or thermoelectric devices. But how we made it is even more promising: Finding new routes to synthesize materials that nature cannot make would catalyze progress towards next-generation technologies."
More information: Sebastian Siol et al. Negative-pressure polymorphs made by heterostructural alloying, Science Advances (2018). DOI: 10.1126/sciadv.aaq1442
Journal information: Science Advances
Provided by SLAC National Accelerator Laboratory



















