Sulfites and bisulfites in shallow lakes may have set the stage for Earth's first biological molecules

Around 4 billion years ago, Earth was an inhospitable place, devoid of oxygen, bursting with volcanic eruptions, and bombarded by asteroids, with no signs of life in even the simplest forms. But somewhere amid this chaotic period, the chemistry of the Earth turned in life's favor, giving rise, however improbably, to the planet's very first organisms.
What prompted this critical turning point? How did living organisms rally in such a volatile world? And what were the chemical reactions that brewed up the first amino acids, proteins, and other building blocks of life? These are some of the questions researchers have puzzled over for decades in trying to piece together the origins of life on Earth.
Now planetary scientists from MIT and the Harvard-Smithsonian Center for Astrophysics have identified key ingredients that were present in large concentrations right around the time when the first organisms appeared on Earth.
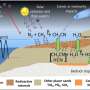
The researchers found that a class of molecules called sulfidic anions may have been abundant in Earth's lakes and rivers. They calculate that, around 3.9 billion years ago, erupting volcanoes emitted huge quantities of sulfur dioxide into the atmosphere, which eventually settled and dissolved in water as sulfidic anions—specifically, sulfites and bisulfites. These molecules likely had a chance to accumulate in shallow waters such as lakes and rivers.
"In shallow lakes, we found these molecules would have been an inevitable part of the environment," says Sukrit Ranjan, a postdoc in MIT's Department of Earth, Atmospheric and Planetary Sciences. "Whether they were integral to the origin of life is something we're trying to work out."
Preliminary work by Ranjan and his collaborators suggest that sulfidic anions would have sped up the chemical reactions required to convert very simple prebiotic molecules into RNA, a genetic building block of life.
"Prior to this work, people had no idea what levels of sulfidic anions were present in natural waters on early Earth; now we know what they were," Ranjan says. "This fundamentally changes our knowledge of early Earth and has had direct impact on laboratory studies of the origin of life."
Ranjan and his colleagues published their results today in the journal Astrobiology.
Setting early Earth's stage
In 2015, chemists from Cambridge University, led by John Sutherland, who is a co-author on the current study, discovered a way to synthesize the precursors to RNA using just hydrogen cyanide, hydrogen sulfide, and ultraviolet light—all ingredients that are thought to have been available on early Earth, before the appearance of the first life forms.
From a chemistry point of view, the researchers' case was convincing: The chemical reactions they carried out in the laboratory overcame longstanding chemical challenges, to successfully yield the genetic building blocks to life. But from a planetary science standpoint, it was unclear whether such ingredients would have been sufficiently abundant to jumpstart the first living organisms.
For instance, comets may have had to rain down continuously to bring enough hydrogen cyanide to Earth's surface. Meanwhile, hydrogen sulfide, which would have been released in huge amounts by volcanic eruptions, would have mostly stayed in the atmosphere, as the molecule is relatively insoluble in water, and therefore would not have had regular opportunities to interact with hydrogen cyanide.
Instead of approaching the origins-of-life puzzle from a chemistry perspective, Ranjan looked at it from a planetary perspective, attempting to identify the actual conditions that might have existed on early Earth, around the time the first organisms appeared.
"The origins-of-life field has traditionally been led by chemists, who try to figure out chemical pathways and see how nature might have operated to give us the origins of life," Ranjan says. "They do a really great job of that. What they don't do in as much detail is, they don't ask what were conditions on early Earth like before life? Could the scenarios they invoke have actually happened? They don't know as much what the stage setting was."
Cranking up the ingredients for life
In August 2016, Ranjan gave a talk at Cambridge University about volcanism on Mars and the types of gases that would have been emitted by such eruptions in the red planet's oxygenless atmosphere. Chemists at the talk realized that the same general conditions would have occurred on Earth prior to the start of life.
"They took away from that [talk] that, on early Earth, you don't have much oxygen, but you do have sulfur dioxide from volcanism," Ranjan recalls. "As a consequence, you should have sulfites. And they said, 'Can you tell us how much of this molecule there would have been?' And that's what we set out to constrain."
To do so, he started with a volcanism model developed previously by Sara Seager, MIT's Class of 1941 Professor of Planetary Sciences, and her former graduate student Renyu Hu.
"They did a study where they asked, 'Suppose you take the Earth and just crank up the amount of volcanism on it. What concentrations of gases do you get in the atmosphere?'" Ranjan says.
He consulted the geological record to determine the amount of volcanism that likely took place around 3.9 billion years ago, around the time the first life forms are thought to have appeared, then looked up the types and concentrations of gases that this amount of volcanism would have produced according to Seager and Hu's calculations.
Next, he wrote a simple aqueous geochemistry model to calculate how much of these gases would have been dissolved in shallow lakes and reservoirs—environments that would have been more conducive to concentrating life-forming reactions, versus vast oceans, where molecules could easily dissipate.
Interestingly, he consulted the literature in a rather unexpected subject while conducting these calculations: winemaking—a science that involves, in part, dissolving sulfur dioxide in water to produce sulfites and bisulfites under oxygenless conditions similar to those on early Earth.
"When we were working on this paper, a lot of the constants and data we pulled out were from the wine chemistry journals, because it's where we have anoxic environments here on modern Earth," Ranjan says. "So we took aspects of wine chemistry and asked: 'Suppose we have x amount of sulfur dioxide. How much of that dissolves in water, and then what does it become?'"
Community cross-talk
Ultimately, he found that, while volcanic eruptions would have spewed huge quantities of both sulfur dioxide and hydrogen sulfide into the atmosphere, it was the former that dissolved more easily in shallow waters, producing large concentrations of sulfidic anions, in the form of sulfites and bisulfites.
"During major volcanic eruptions, you might have had up to millimolar levels of these compounds, which is about laboratory-level concentrations of these molecules, in the lakes," Ranjan says. "That is a titanic amount."
The new results point to sulfites and bisulfites as a new class of molecules—ones that were actually available on early Earth—that chemists can now test in the lab, to see whether they can synthesize from these molecules the precursors for life.
Early experiments led by Ranjan's colleagues suggest that sulfites and bisulfites may have indeed encouraged biomolecules to form. The team carried out chemical reactions to synthesize ribonucleotides with sulfites and bisulfites, versus with hydrosulfide, and found the former were able to produce ribonucleotides and related molecules 10 times faster than the latter, and at higher yields. More work is needed to confirm whether sulfidic anions were indeed early ingredients in brewing up the first life forms, but there is now little doubt that these molecules were part of the prebiotic milieu.
For now, Ranjan says the results open up new opportunities for collaboration.
"This demonstrates a need for people in the planetary science community and origins-of-life community to talk to each other," Ranjan says. "It's an example of how cross-pollination between disciplines can really yield simple but robust and important insights."
More information: Sukrit Ranjan et al. Sulfidic Anion Concentrations on Early Earth for Surficial Origins-of-Life Chemistry, Astrobiology (2018). DOI: 10.1089/ast.2017.1770
Journal information: Astrobiology
Provided by Massachusetts Institute of Technology
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.