January 30, 2018 feature
Did water-based life originate without water?
When trying to understand the origins of life on Earth, researchers run into a paradox: while water is an indispensable solvent for all known life forms that exist today, water also inhibits the formation of string-like chains of nucleic acid polymers such as RNA that were likely precursors of life. This raises the question: how could the nucleic acids have formed in the first place? One solution to this "water paradox" is that life may have originated in something other than water, and only later adapted to the presence of water.
"We are fascinated by the possibility that water-based life may have originated without water at all," Zachary Adam, a researcher at Harvard University, told Phys.org.
Adam and others have been investigating a leading candidate for a water alternative called formamide, a clear liquid that consists of hydrogen, oxygen, carbon, and nitrogen. Not only does formamide favor polymer bond formation more than water does, it also reacts with other molecules to form nucleobases, amino acids, and some of the other basic compounds needed to make nucleic acids.
But there is a glaring problem with this proposal: formamide does not occur naturally in any significant quantity anywhere on Earth. Although formamide is widely used in industry as a solvent for making pharmaceuticals and pesticides, all of this formamide is synthetically produced.
Formamide does exist in space, however, which has previously motivated researchers to suggest that it may have been transported to Earth via comets or meteors. But it is unlikely that this scenario could have produced the large, concentrated reservoirs of formamide needed for life's precursors to form.
Now in a new paper published in Scientific Reports, a team of researchers, led by Adam and coauthor Masashi Aono at Keio University and Tokyo Institute of Technology, have demonstrated the possibility that formamide may have been produced in abundance by radiation in some pockets of the early Earth.
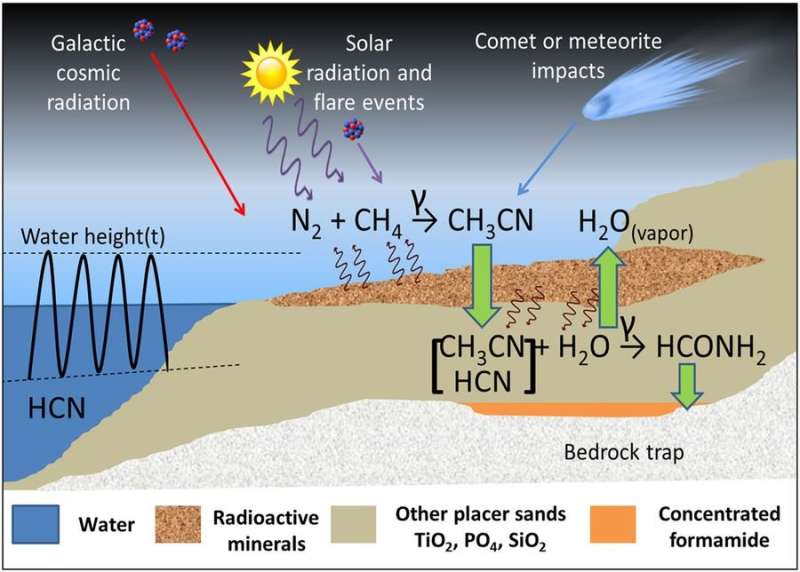
In experiments, the researchers irradiated hydrogen cyanide and acetonitrile—two chemicals present on early Earth—with gamma rays. They found that formamide was one of the major products.
Although in their experiments the researchers used a cylinder of cobalt-60 to produce the gamma rays, they suggest that on early Earth the radiation may have come from radioactive mineral deposits (found today on beaches worldwide) or uranium fission zones. Only one region is currently known to contain evidence of a small handful of uranium zones that existed in Earth's geologic history—the Oklo region in Gabon, Africa—but these zones only became active long after life originated.
The researchers calculated that, if similar zones existed 4 billion years ago, a single site could have produced over 6 orders of magnitude more formamide over a given area than that estimated by delivery from comets and meteorites. The results suggest that radioactive mineral deposits can produce enough formamide to accumulate to high concentrations, which could have formed large formamide reservoirs in which nucleic acids could have formed as precursors to the first living organisms.
"Often the problem of the origins of life is considered to be solved if we could understand how prototypical building blocks of life such as biopolymers and metabolites could form in plausible early-Earth environments," Aono said. "But we are not satisfied with this way of thinking. Life should not be treated as a bag full of the building blocks, but should be understood as a complex network of chemical reactions."
As the researchers explain, radiation is particularly unique as an energy source for the origins of life compared to redox chemistry or simple heating. As Adam said, this is because radiation "drives an expansive network of reactions, not just an array of products for an array of inputs."
Of course, the researchers have only showed what could have happened, and not what did happen. In the future, they plan to continue studying all of the possible scenarios for the origins of life and examine the plausibility of each occurring, and see where the evidence leads.
"We are now trying to assess whether the full network of the driven reactions exhibits attributes found across many different scales of complex living systems, such as cellular metabolic networks, population dynamics, and even ecological relationships," Aono said.
More information: Zachary R. Adam et al. "Estimating the capacity for production of formamide by radioactive minerals on the prebiotic Earth." Scientific Reports. DOI: 10.1038/s41598-017-18483-8
Zachary R. Adam et al. "Subsumed complexity: abiogenesis as a by-product of complex energy transduction" Philosophical Transactions of The Royal Society A. DOI: 10.1098/rsta.2016.0348
Related video: Zachary Adam's lecture on "Energy, Entropy, and Complexity on the Prebiotic Earth"
Journal information: Scientific Reports , Philosophical Transactions of the Royal Society A
© 2018 Phys.org