The 'radical' ways sunlight builds bigger molecules in the atmosphere
With summer approaching, "sea and sun" might conjure up images of a beach trip. But for scientists, the interactions of the two have big implications for the climate and for the formation of tiny droplets, or aerosols, that lead to clouds. In ACS Central Science, researchers demonstrate that sunlight can cause certain molecules at the ocean's surface to activate others, resulting in larger molecules that could affect the atmosphere.
Certain organic molecules become activated and react when they absorb sunlight. They often go through a reactive intermediate called a "radical," which initiates a chain reaction leading to the formation of more complex chemicals. This "radical initiator" pathway is important for understanding which molecules at the sea surface end up in the atmosphere, where they seed clouds. Which molecules are found in the atmosphere on aerosols will determine whether they absorb or reflect sunlight, affecting the temperature of the planet. Until now, scientists had focused much of their attention on the hydroxyl radical, which reacts very efficiently in the atmosphere. Rebecca Rapf, Veronica Vaida and colleagues at the University of Colorado propose that a class of compounds called α-keto acids can be photo-activated by sunlight and drive reactions with molecules that do not themselves absorb sunlight.
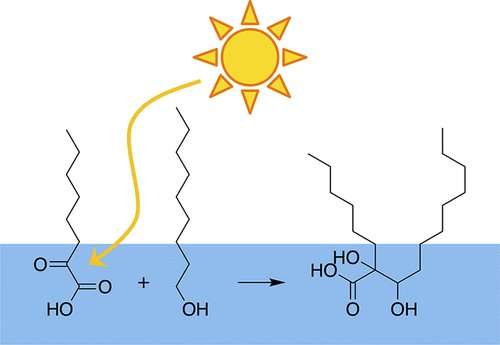
The researchers studied two different α-keto acids and showed that light caused the acid to react with several fatty acids and alcohols. These classes of molecules are commonly found near the ocean's surface, and they are ubiquitous in biology. The authors explain that this sunlight-initiated chemistry could change the composition of the sea surface. The new, larger molecules formed may add to aerosols, changing their properties and leading to interesting and previously unforeseen consequences to human health, visibility and climate.
More information: Rebecca J. Rapf et al. Environmental Processing of Lipids Driven by Aqueous Photochemistry of α-Keto Acids, ACS Central Science (2018). DOI: 10.1021/acscentsci.8b00124
Abstract
Sunlight can initiate photochemical reactions of organic molecules though direct photolysis, photosensitization, and indirect processes, often leading to complex radical chemistry that can increase molecular complexity in the environment. α-Keto acids act as photoinitiators for organic species that are not themselves photoactive. Here, we demonstrate this capability through the reaction of two α-keto acids, pyruvic acid and 2-oxooctanoic acid, with a series of fatty acids and fatty alcohols. We show for five different cases that a cross-product between the photoinitiated α-keto acid and non-photoactive species is formed during photolysis in aqueous solution. Fatty acids and alcohols are relatively unreactive species, which suggests that α-keto acids are able to act as radical initiators for many atmospherically relevant molecules found in the sea surface microlayer and on atmospheric aerosol particles.
Journal information: ACS Central Science
Provided by American Chemical Society