Researchers discover how colliding oxygen molecules absorb light
Scientists at Radboud University have managed to do what had never been tried before: accurately describe how colliding oxygen molecules absorb light. Our atmosphere consists of approximately 20 percent oxygen molecules, which constantly collide with each other and with the 80 percent of nitrogen molecules, and in doing so absorb light. The new theory describes the mechanism by which this takes place. This discovery will enable climate researchers to far more accurately determine the concentrations of the molecules that pollute the atmosphere and contribute to the greenhouse effect.
With the aid of satellites in space, we are able to accurately measure the main pollutants and greenhouse gases in the atmosphere. In order to do so, the satellites observe the spectrum of sunlight that shines through the atmosphere. Molecules of various atmospheric gases absorb sunlight, and this absorption occurs at different wavelengths for each gas. Since the 'fingerprint' (the spectrum) of each of the different gases is known, scientists now know more about the presence and concentration of each of these gases.
To correct these measurements for the effects of clouds and changes in air pressure, the absorption by oxygen is measured as a reference, because we know exactly how much oxygen is present in the atmosphere. Scientists at Radboud University now show that the 'fingerprint' of oxygen is different from what it was believed to be, because it is strongly influenced by collisions with nitrogen and collisions with other oxygen molecules.
Light absorption due to collision
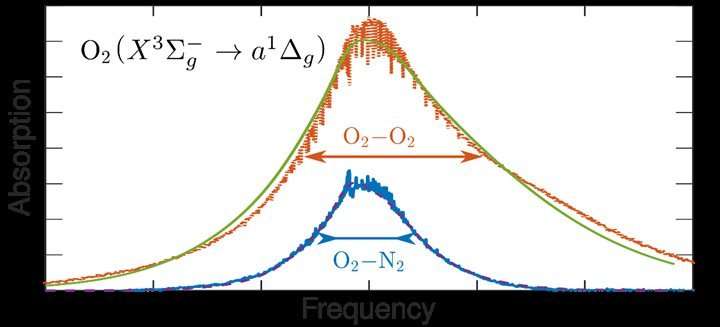
Oxygen, in the form of O2 molecules, is, after nitrogen (N2), the most commonly occurring gas in the atmosphere. Stable molecules are almost never magnetic, but oxygen is. When an oxygen or nitrogen molecule collides with another particle, there is a shift of the electrical charges in the molecule. A dipole is created that works as an antenna, enabling the molecule to absorb light. The theory that has now been developed shows that, contrary to expectations, the effect of collisions of oxygen with other oxygen molecules is very different from the effect of collisions with nitrogen molecules. It is shown that the reason for this difference is that oxygen is magnetic whereas nitrogen is not.
Measuring absorption
The scientists examined this initially with an experiment: In a tank with oxygen gas, they measured the spectrum of light absorption by oxygen molecules at different pressures. If the pressure increases, the molecules collide more frequently with each other resulting in more light absorption, but also the shape of the absorption spectrum changes. In order to understand this, the scientists developed a complicated new theory for the absorption of light by colliding oxygen molecules. With the aid of this theory, the satellite measurements of the atmosphere can be interpreted more accurately.
It seemed impossible…
It was by no means certain that developing a good model for light absorption by colliding oxygen molecules would be successful. Professor of Theoretical Chemistry Gerrit Groenenboom: "Some scientists said that we'd never be able to do it. And when we started, it seemed as if no method whatsoever could provide reliable results. Ultimately, with the use of a newly developed method, we managed to show that, in spite of a certain degree of uncertainty, the shape of the spectrum can be accurately predicted." It was also shown that this shape can be described with an analytical function, which is convenient for future applications.
Same experiment, different result
Also astronomers have been helped by the scientists' findings. Scientists who examine the atmosphere use the HITRAN Database of the Harvard-Smithsonian Center for Astrophysics, which contains the spectra of a large collection of molecules. However, measurements of the oxygen spectrum by two different research groups provided very different results. It was not clear which of the results was sufficiently reliable to be included in the database. This problem could be solved with the aid of the theoretical model developed in Nijmegen: the line shape resulting from one of the two measurements was shown to differ fundamentally from the theory.
This does not mean that the experiment itself was incorrect, but rather the interpretation of it. Researcher Tijs Karman: "Astronomers also use this database to interpret spectra from the atmosphere of exoplanets and, for example, to see if oxygen is present, which is regarded as a sign of life."
More information: O2-O2 and O2-N2 collision-induced absorption mechanisms unraveled. Nature Chemistry, 2018, DOI: 10.1038/s41557-018-0015-x
Journal information: Nature Chemistry
Provided by Radboud University



















