Water without windows: Capturing water vapor inside an electron microscope

The inside of an electron microscope, which requires vacuum levels similar to those encountered in outer space, can be an extremely inhospitable place for organic materials. Traditionally, life scientists have circumvented this problem by freezing their specimens so that that they can be safely loaded into a microscope. Now, researchers at the Okinawa Institute of Science and Technology Graduate University (OIST) have devised a new approach to imaging organic compounds.
By suspending organic samples in water vapor, OIST scientists were able to demonstrate another way to view them at high resolution. The researchers found they could send an electron beam, commonly used in microscopy, through vapor dense enough that it might be possible to keep samples in their native, wet state and still allow for ultrahigh resolution imaging.
Their study, published in the journal PLOS ONE, applies physics to a well-known problem in biology. The results could simplify what is currently a difficult process of imaging organic materials.
Usually, in order to view samples—particularly, fragile organic samples—inside a high-powered transmission electron microscope, scientists must undertake extensive preparation. Creating a plate of ice a fraction of a nanometer thick with a particular crystal structure can require many trials. This labor-intensive process, which can take months, inspired Cathal Cassidy, lead author on the paper and a researcher at OIST's Quantum Wave Microscopy Unit, to try another method.
"I saw my colleagues investing a lot of effort in this," said Cassidy, "and I thought, 'Couldn't we just avoid this ice thing altogether?'"
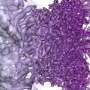
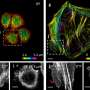
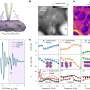
The researchers first used gold, an inorganic material, to demonstrate that atoms can be successfully imaged inside water vapor. Then, they looked at a virus using the same method. The sample remained stable, and the resulting image came out crisp, in relatively high resolution.
The researchers' method eliminates the need to freeze a sample or view it through a chamber. Although effective, each of these commonly used methods comes with disadvantages.

Ideally, the ice acts as a clean slate, or a window—relatively translucent, it allows scientists to view the materials suspended inside it with minimal interference. Hailed for "bringing biochemistry into a new era" by the Swedish Academy, this method received the 2017 Nobel Prize in chemistry. However, freezing does not allow scientists to study dynamic processes—like the live interaction of a virus with a host cell.
Alternately, scientists can view organic samples by suspending them in liquid, enclosed in a chamber with ultra-thin windows. These windows prevent the liquid from seeping into the vacuum chamber and damaging the electron gun. Yet, thin as they are, even these minimal barriers degrade image quality. The chamber's geometry also significantly limits scientists in how much they can tilt a sample for a three-dimensional view.
The method devised by OIST researchers provides a feasible alternative to these popular approaches. The sample is suspended in water vapor, which is pumped into the portion of the tube surrounding the sample and rapidly pumped out again. Tiny apertures above and below the sample allow the electron beam to pass directly through it. Because the sample is not enclosed by ice or glass, it can be tilted for three-dimensional imaging.

Cassidy emphasized that the study is a first step toward high-resolution imaging of hydrated samples in water vapor. He said he hoped biologists would build on the results. The researchers' study and additional materials—including raw data—can be found in the journal PLOS ONE.
"Anybody who wants to try it or play with it, they can do it," he said, pointing to the availability of data. "If somebody else takes the baton and pushes this forward, I'd be really happy with that."
More information: Cathal Cassidy et al. Water without windows: Evaluating the performance of open cell transmission electron microscopy under saturated water vapor conditions, and assessing its potential for microscopy of hydrated biological specimens, PLOS ONE (2017). DOI: 10.1371/journal.pone.0186899
Journal information: PLoS ONE
Provided by Okinawa Institute of Science and Technology