From the omelette to the egg: Reversing protein aggregations

To cook an omelette, you have to scramble an egg, and like Humpty Dumpty it can never be put back together again. This is because the egg undergoes a set of physiological and chemical changes as it cooks, which cause its chemical bonds to break and its proteins to aggregate, restructuring and setting into a new, final—and irreversible—shape.
However, a new Tel Aviv University study suggests for the first time a novel form of protein aggregation that is both reversible and has positive physiological consequences for cells. The discovery may eventually lead to new therapies for neurodegenerative conditions such as Alzheimer's, Parkinson's and "mad cow" diseases.
The research study was led by Prof. Martin Kupiec and conducted by Dr. Kobi Simpson-Lavy, both of TAU's School of Molecular Cell Biology and Biotechnology, and published in the journal Molecular Cell.
How to "de-blob" a protein
"Most of the functions within our cells are carried out by proteins. But when these proteins aggregate, they produce a 'blob' that renders them inactive," Prof. Kupiec says. "Protein aggregation tends to increase with age and leads to a number of human diseases, particularly those resulting in neurodegeneration."
"Moreover, when proteins adopt an erroneous configuration—when they're misfolded—the cells attempt to take the clumps apart, or to pile them up at particular locations in the cell, to minimize their toxic effect," Dr. Simpson-Lavy says. "This process has been linked to the development of a number of neurodegenerative conditions, such as Alzheimer's, Parkinson's and 'mad cow' diseases."
The new study examines an entirely different type of protein aggregation, which may provide a new mechanism with which to regulate the activity of genes according to changes in the cell's environment.
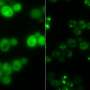
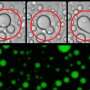
The research for the study emerged serendipitously. While Dr. Simpson-Lavy was studying the metabolism of sugars in yeast cells, he noticed that a tagged version of the protein he was observing—Std1—formed a bright splotch outside the cell's nucleus whenever glucose was added to the cells.
In other words, Std1, which is usually present in the cells' nuclei, where the genome resides, ended up in an aggregation outside of the nuclei.
"We wondered whether this protein aggregation would change according to different conditions in the microenvironment of the cell," Prof. Kupiec says. "And indeed, when conditions changed again and glucose was depleted, the aggregate dissolved and the Std1 protein could be seen again in the nucleus. Std1 plays a role in responding to different sugars in the growth medium, so the reversible aggregation and dissolution of Std1 allowed the cell to respond quickly to varying levels of sugar abundance."
Bad—and good—for you
The study also suggests that not all protein aggregates are "bad for you." Some play important physiological and regulatory roles. According to the study, the "molecular chaperones" that have been found to fuel many neurodegenerative diseases may have originally been intended to regulate the buildup of non-pathological proteins.
"These results could open the way for possible future treatments that may try to change the aggregation from irreversible to reversible," Prof. Kupiec says. "If we can find out how to turn an irreversible aggregation into a reversible one, it would be possible to treat neurodegenerative diseases and reverse the effect of the aggregates.
"In other words, it may still be possible to reconstruct an egg from an omelette," Prof. Kupiec observes.
The researchers are currently examining what makes the same protein behave differently under different conditions.
More information: Kobi Simpson-Lavy et al, The Std1 Activator of the Snf1/AMPK Kinase Controls Glucose Response in Yeast by a Regulated Protein Aggregation, Molecular Cell (2017). DOI: 10.1016/j.molcel.2017.11.016
Journal information: Molecular Cell
Provided by Tel Aviv University