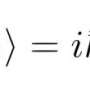Novel computational approach launches new paradigm in electronic structure theory

A group of Michigan State University (MSU) researchers specializing in quantum calculations has proposed a radically new computational approach to solving the complex many-particle Schrödinger equation that holds the key to explaining the motion of electrons in atoms and molecules.
By understanding the details of this motion, one can determine the amount of energy needed to transform reactants into products in a chemical reaction, or the color of light absorbed by a molecule, and ultimately accelerate the design of new drugs and materials, better catalysts and more efficient energy sources.
The work, led by Piotr Piecuch, a University Distinguished Professor in the MSU Department of Chemistry and an adjunct professor in the Department of Physics and Astronomy in the College of Natural Science, was published in the Dec. 1 issue of Physical Review Letters. Also involved in the work are fourth-year graduate student J. Emiliano Deustua and senior postdoctoral associate Jun Shen. The group provides details for a new way of obtaining highly accurate electronic energies by merging the deterministic coupled-cluster and stochastic Quantum Monte Carlo approaches.
Although the general mathematical theory of the microscopic world of quantum mechanics is well established, the challenges have been centered on solving the complicated equations born of the exact application of the laws. The many-particle Schrödinger equation is at the heart of the problem.
"Instead of insisting on a single philosophy when solving the electronic Schrödinger equation, which has historically been either deterministic or stochastic, we have chosen a third way," Piecuch said. "As one of the reviewers noted, the essence of it is remarkably simple: use the stochastic approach to determine what is important and the deterministic approach to determine the important, while correcting for the information missed by stochastic sampling."
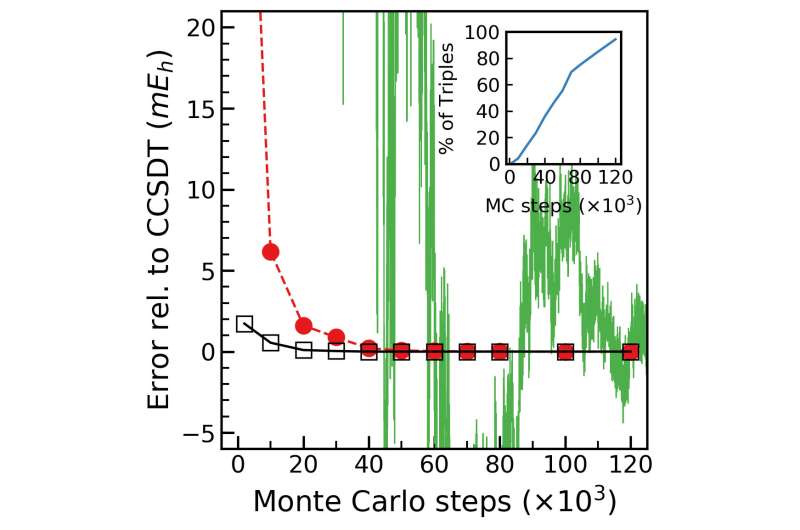
Their new method displays rapid convergence toward target molecular electronic energetics based on the information extracted from the early stages of the Monte Carlo wave function propagations, reducing computational costs by orders of magnitude.
Solving the Schrödinger equation for the many-electron wave function has been a key challenge in quantum chemistry for decades. Anything other than a one-electron problem, such as a hydrogen atom, requires resorting to numerical methods, converted into sophisticated computer programs, such as those developed by Piecuch and his group. The main difficulty has been the intrinsic complexity of the electronic motion, which quantum chemists and physicists call "electron correlation."
The new idea is to use the stochastic methods, developed by Ali Alavi, professor of chemistry at the University of Cambridge and director of Electronic Structure Theory Group in the Max Planck Institute for Solid State Research in Stuttgart; George H. Booth, Royal Society Research Fellow in King's College London; and Alex J. W. Thom, Royal Society Research Fellow in the University of Cambridge, to identify the leading wave function components and the deterministic coupled-cluster computations, combined with suitable energy corrections, to provide the missing information.
"It's like playing chess and being able to predict the outcome of the game after the initial few moves," Deustua said.
The findings may profoundly impact quantum calculations for atoms and molecules, and other many-electron systems.
The merging of deterministic and stochastic approaches as a general method of solving the many-particle Schrödinger equation may also impact other areas, such as nuclear physics.
"In the case of nuclei, instead of being concerned with electrons, one would use our new approach to solve the Schrödinger equation for protons and neutrons," Piecuch said. "The mathematical and computational issues are similar. Just like chemists want to understand the electronic structure of a molecule, nuclear physicists want to unravel the structure of the atomic nucleus. Once again, solving the many-particle Schrödinger equation holds the key."
More information: J. Emiliano Deustua et al. Converging High-Level Coupled-Cluster Energetics by Monte Carlo Sampling and Moment Expansions, Physical Review Letters (2017). DOI: 10.1103/PhysRevLett.119.223003
Journal information: Physical Review Letters
Provided by Michigan State University