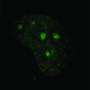
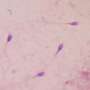
Endogenous protein reverses scarring in the lungs
The endogenous protein RAGE, which has usually been negatively associated with chronic inflammation and diabetic complications, plays a major role in the repair of DNA damage—and also appears to heal tissue damaged as a result of accelerated cell senescence. This molecular mechanism was described in a study by scientists from Heidelberg University Hospital and the German Center for Diabetes Research, which was publshed In the October issue of the journal Nucleic Acids Research. They discovered the potential therapeutic benefit of the protein in mice that are unable to produce RAGE. As a result of the limited DNA repair, they develop pronounced pulmonary fibrosis, i.e. scarring in the lungs. After treatment with the protein, the scarring healed.
"This is astonishing in that fibrosis has so far been considered irreversible. With RAGE, we could, for the first time, have found a possible starting point to cure this frequent tissue damage," said senior author Professor Peter Nawroth, medical director of the Department of Endocrinology, Metabolism and Clinical Chemistry at Heidelberg University Hospital. "Many questions—e. g. how this healing works in detail—are still unanswered." The project was funded by the Dietmar Hopp Foundation and subsequently by the German Center for Diabetes Research.
RAGE (Receptor of Advanced Glycation Endproducts) is well known in medical research. The protein plays a decisive role not only in diabetes but also in chronic and excessive inflammatory reactions such as atherosclerosis and sepsis, but also in Alzheimer's disease and cancer development. The protein is mainly active on the surfaces of tissue cells and cells of the immune system. On the other hand, inside the cell nucleus, RAGE is responsible for the error-free repair of severe DNA damage, known as double-strand breaks, the Heidelberg researchers discovered. In these cases of damage, the two interconnected and twisted strands of DNA are completely cut off. Without immediate repair, the cell would quickly perish.
Since the DNA in the cells is continuously "read"—it contains the blueprint for all processes in the cell—and is thus in constant use, damage is extremely frequent. Seen purely mathematically, in each body cell, defects in this vital molecule probably occur several thousand times a day. Disturbances in the complex repair mechanisms, for example because the cells are particularly exposed to toxins from the environment or harmful metabolic products, can lead to rapid aging, degeneration and scarring of entire tissue groups. Examples are liver fibrosis caused by alcohol abuse or retinal and kidney damage caused by diabetes mellitus. At present, there are no effective drugs to specifically remedy disorders of these repair mechanisms and prevent tissue damage, e.g. in the case of diabetes.
Mice that are unable to form RAGE due to a genetic defect will develop pulmonary fibrosis. The lungs are particularly susceptible to tissue damage, as they are in constant contact with the outside world through the air they breathe and are particularly exposed to environmental influences. In the animal model, the researchers succeeded in elucidating the hitherto unknown molecular mechanism of DNA repair under RAGE involvement and in identifying further important protagonists. If they introduced RAGE into the mice's lungs with the help of modified viruses, it was not only DNA repair that normalized—to the scientists' surprise, the scarred tissue regenerated and regained some of its functionality.
The published study not only provides important insights into the molecular relationship between RAGE-mediated DNA repair, cell aging and fibrosis. For the first time ever, molecular therapy for the repair of damage to the lungs caused by genes and cells and thus for the prevention of fibrosis or tumors, which also occur as a result of DNA damage, may be within reach," said first author Dr. Varum Kumar, Department of Endocrinology, Metabolism and Clinical Chemistry at Heidelberg University Hospital and the German Center for Diabetes Research. Next, the scientists want to investigate whether RAGE also plays a role in liver and kidney fibrosis and whether treatment with the protein can also correct damage to these organs. The team has already patented the RAGE therapy using modified viruses.
More information: Varun Kumar et al, Homeostatic nuclear RAGE–ATM interaction is essential for efficient DNA repair, Nucleic Acids Research (2017). DOI: 10.1093/nar/gkx705
Journal information: Nucleic Acids Research
Provided by Deutsches Zentrum fuer Diabetesforschung DZD