A molecular garbage disposal complex has a role in packing the genome
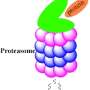
New research from the Korea Institute of Science and Technology, to be published in the Journal of Biological Chemistry on Oct. 13, has found that the proteasome, an essential protein complex that breaks down proteins in cells, has another unexpected function: directly regulating the packing of DNA in the nucleus.
The proteasome breaks down proteins that the cell has tagged for degradation in a process called proteolysis. Dysfunction in the proteasome has been observed in diseases of many physiological systems, from the immune, nervous and cardiovascular systems to the whole organism's aging processes. Increasing research suggests that, like a Swiss army knife with hidden tools, the proteasome is able to perform additional functions that don't involve proteolysis.
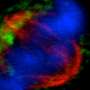
DNA is organized in the nucleus in complexes with protein in the nucleus in a form called chromatin. Broadly speaking, loosely packed chromatin, or euchromatin, allows DNA to be transcribed and genes to be expressed, whereas tightly packed heterochromatin prevents gene expression.
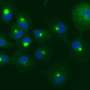
In experiments using yeast cells, Hogyu David Seo, a graduate student in Daeyoup Lee's lab, found that the proteasome could induce heterochromatin to form in some parts of the genome but stop it from spreading to other regions. Surprisingly, the mutations in the proteasome that revealed the proteasome's effects on chromatin had no effect on proteolysis, meaning that the proteasome affects heterochromatin through an activity other than proteolysis. How it does this is not yet known.
"[The proteasome] can exert force on proteins and translocate, tilt, bend them," Seo said. "So I believe the proteasome physically modulates proteins that act as a shield for heterochromatin. That's how I think it might work."
Heterochromatin formation and spread is of interest in the field of epigenetics, because changes in the chromatin state of cells in one generation can potentially be passed on to the next generation.
"[The proteasome] may have some effect on epigenetic programming inheritance because it affects the spreading of heterochromatin," Seo said. "I'm not really sure how it might work, because there are so many ways that it could act, but I'm sure that it may exert some effects on epigenetic programming."
For now, the team is focusing on understanding how the proteasome regulates heterochromatin in organisms besides yeast, including mice and human cells.
"The proteasome engages with virtually every protein in our body with respect to the protein-degradation function," Lee said. "We believe that our work is just a glimpse of what this protein can do...Dissecting the proteasome functions will definitely help to develop therapeutic strategies to various diseases, such as neurological diseases and cancer."
More information: Hogyu David Seo et al, The 19S proteasome is directly involved in the regulation of heterochromatin spreading in fission yeast, Journal of Biological Chemistry (2017). DOI: 10.1074/jbc.M117.790824
Journal information: Journal of Biological Chemistry