'Hypermutators' drive pathogenic fungi to evolve more rapidly

Mutations tend to get a bad rap, and deservedly so. A single defect in our DNA can strip us of our sight, thicken our lungs with mucus, prompt us to bleed to death, weaken our muscles or fill our organs with tumors.
But in certain situations, a mutation can actually be a source of strength. For microorganisms, the right mutation can confer superpowers, allowing them to traverse continents, infect new hosts and avoid drug-induced annihilation.
Microbes are such big fans of mutations that they have rigged their genomes to accumulate more and more of them.
In a study published September 26 in eLife, Duke researchers show that lineages of the fungal pathogen Cryptococcus deuterogattii house a specific mutation in their DNA that increases their mutation rate. These 'hypermutators,' as they are called, rapidly develop resistance to the antifungal drugs FK506 and rapamycin.
"If there weren't mutations, there wouldn't be any raw genetic material for evolution and selection to act upon," said Joseph Heitman, senior study author and professor and chair of molecular genetics and microbiology at Duke University School of Medicine. "These hypermutators are probably a lot more common that we think, particularly among pathogenic fungi."
For nearly two decades, a rare but potentially deadly fungus called Cryptococcus deuterogattii has gained a foothold in the Pacific Northwest and Vancouver Island. Unlike other fungi that predominantly infect patients with compromised immune systems, Cryptococcus deuterogattii has sickened hundreds of otherwise healthy people. The emerging fungal pathogen causes severe pulmonary and central nervous system infections, and is fatal if left untreated.
Blake Billmyre, a former graduate student in Heitman's lab, sequenced the entire genome of three different strains of C. deuterogattii (a clinical isolate from Seattle in the early 1970s, an environmental isolate from a eucalyptus tree in the San Francisco area circa 1990, and a clinical isolate from Brazil from 1980) that are most closely related to those causing the ongoing outbreak. He discovered that all three isolates of the fungus were missing a single base of DNA in MSH2, one of several genes involved in "mismatch repair" to fix mistakes that arise during DNA replication.
Interestingly, humans that harbor mutations in MSH2 have a condition known as Lynch syndrome. Because their genomes accumulate errors more quickly than most people, these patients carry a 75 percent lifetime risk of developing colorectal cancer, and are also more likely to develop cancers of the urinary tract, ovary, stomach, small intestine, hepatobiliary tract, skin and brain.
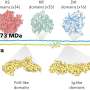
Billmyre decided to test whether mutations in MSH2 in fungi also caused their genomes to mutate at a faster pace. He and Shelly Clancey, a technician in Heitman's lab, set up competition cultures of Cryptococcus deuterogattii, pitting wildtype strains with those containing an MSH2 hypermutator mutation. When they grew the fungi under standard, non-stress conditions, the strains grew in test tubes at the same rate. However, when they grew them under stressful conditions, adding the selective pressure of antifungals like rapamycin and FK506, the hypermutator strains took over almost all of the cultures because they could more rapidly acquire resistance to the drugs.
By accumulating mutations, the fungi were able to survive drug treatment. But in the process, they lost much of their virulence. The researchers found that all three strains with the hypermutator were significantly weakened compared to the outbreak strains. "We think that is because of the thousands of mutations caused by the hypermutator," said Heitman. "It seems to cause a mutational meltdown."
The researchers are currently looking to see what percentage of other clinical isolates are hypermutators. Their findings thus far indicate it could be a significant number.
"Multiple studies have come out recently indicating that hypermutators may be present in bacteria, fungi and perhaps even higher eukaryotic populations. So it is likely to be pretty widespread and is an important consideration in terms of thinking about how antifungal drug resistance develops," said Billmyre, lead study author and now a postdoctoral fellow at the Stowers Institute for Medical Research.
More information: R Blake Billmyre et al, Natural mismatch repair mutations mediate phenotypic diversity and drug resistance in Cryptococcus deuterogattii, eLife (2017). DOI: 10.7554/eLife.28802
Journal information: eLife
Provided by Duke University