Fungal sex can generate new drug resistant, virulent strains
Though some might disagree, most biologists think the purpose of sex is to create diversity among offspring. Such diversity underpins evolution, enabling organisms to acquire new combinations of traits to adapt to their environment.
However, scientists have been perplexed to find that many fungi and microorganisms procreate with exact replicas of themselves, where the expected outcome would simply be more of the same.
Now researchers have found the act of sex between such genetically identical organisms can itself be mutagenic, meaning it can create genetic changes and diversity where it did not previously exist. Studies of a fungus called Cryptococcus showed the process of sexual reproduction can result in extra bundles of genetic material or chromosomes that can be beneficial to the organism's survival.
The discovery not only contributes to the understanding of sex, but also lends insight into how pathogenic microbes can evolve to cause and spread diseases, such as the ongoing outbreak of Cryptococcus in Canada and the Pacific Northwest of the United States. The results were published Sept. 10, 2013, in the journal PLOS Biology.
"These studies turn our view of the functions of sex by 180 degrees and reveal that sex doesn't just mix up already existing genetic diversity, but can actually create it from scratch," said Joseph Heitman, M.D., Ph.D., the study's senior author and professor and chair of molecular genetics and microbiology at the Duke University School of Medicine.
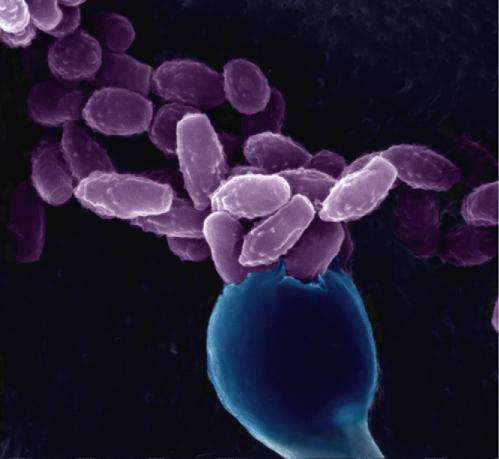
The fungi Cryptococcus neoformans is a global pathogen that primarily infects individuals with compromised immune systems, such as HIV/AIDS patients. It causes more than 600,000 deaths a year from cryptococcal pneumonia and meningoencephalitis, accounting for a third of all AIDS-related deaths.
Researchers have been interested in the sexual cycle of Cryptococcus because it is the only way this fungal pathogen can produce spores to infect its host. Though it has two mating types—namely, "a" and "alpha"—the vast majority of the fungi found in nature are of the alpha variety. Many had assumed this finding meant the fungi had given up on finding mates and was resorting to asexual reproduction. That is, until a previous study by Heitman and his colleague and former fellow Xiaorong Lin showed that Cryptococcus engaged in a phenomenon known as unisexual reproduction, or sex involving fungi of the same mating type.
"That was a big mystery because if there are two identical genomes coming together, the end product should be the same as if the fungi had just cloned itself through asexual reproduction," said Heitman. "We were trying to get a sense of, is it just spinning its wheels, or maybe there is some other function to it."
Heitman hypothesized that sex might be a means of creating genetic diversity. To test this hypothesis, he and his colleagues, former fellow Min Ni and current graduate student Marianna Feretzaki, grew the microbe in two different ways—one where it underwent asexual reproduction, the other where it underwent unisexual reproduction. They then took the offspring of those reproductive cycles and compared them to each other and to the starting parent.
The researchers found the offspring of asexual reproduction were essentially clones, looking just like each other and just like the parent. On the other hand, some of the offspring of unisexual reproduction differed, both in terms of genetic makeup and behavior. Those differences were due to a distinct genetic change called aneuploidy, or having more than the normal number of DNA-packaging chromosomes.
Aneuploidy is well known to be deleterious in humans, causing genetic disorders like Down's syndrome or trisomy 21. But having an extra chromosome can actually be beneficial in microbes like Cryptococcus, where it has been shown to confer drug resistance to the antifungal fluconazole.
In this study, the researchers discovered that aneuploidy could lead to beneficial, neutral or detrimental changes in the fitness of the offspring. For example, offspring with an extra copy of chromosome 9 or 10 became drug resistant and were able to outcompete their parent when subjected to antifungal treatment.
Heitman and his colleagues also used some of the aneuploid offspring to infect animal models and showed they could be just as virulent as the parent, suggesting how new properties (drug resistance) and old ones (virulence) could be combined.
"An interesting feature of aneuploidy is it can be temporary," said Heitman. "If at some point it stops being beneficial or adaptive, the fungi can lose the chromosome just as easily as they gained it. In fact, we showed that if we took the aneuploid offspring and let them lose their extra chromosomes, they also lose their new properties and go back to behaving just like their parent did."
The key finding, he said, "illustrates how unisexual reproduction introduces limited genetic diversity in clonal populations already well adapted to an environment, which may drive outbreaks of drug resistant pathogenic microbes."
Heitman is currently looking for aneuploidy in samples from the outbreak of Cryptococcus gattii in the western part of North America to see if it can explain how this sister strain of Cryptococcus neoformans has come to infect otherwise healthy individuals.
More information: "Unisexual and Heterosexual Meiotic Reproduction Generate Aneuploidy and Phenotypic Diversity de novo in the Yeast Cryptococcus neoformans," Min Ni, Marianna Feretzaki, Wenjun Li, Anna Floyd-Averette, Piotr Mieczkowski, Fred S. Dietrich, Joseph Heitman. PLOS Biology, Sept. 10, 2013.
Journal information: PLoS Biology
Provided by Duke University