Controlling traffic on the electron highway—researching graphene

On an otherwise normal day in the lab, Eva Andrei didn't expect to make a major discovery. Andrei, a physics professor at Rutgers University, was using graphite – the material in pencils – to calibrate a scanning tunneling microscope. As part of the process, she turned on a very powerful magnetic field. When she looked up to see the material's electronic spectrum, she was astonished. "We saw huge, beautiful peaks up there, just incredible. And they didn't make any sense," she recalled.
Remembering a lecture she'd recently attended, she realized the graphite had separated out into sheets just one atom thick. This material, known as graphene, has bizarre electronic properties. But even for graphene, the spectrum she saw was strange. In fact, no one had ever seen anything like it before. As Andrei described it, her colleague "went berserk in the corridor and just yelled 'Graphene!'" Andrei had made a serendipitous discovery – a new electric phenomenon.
This was neither the first nor last time that electrons' movement in graphene would surprise and elate scientists. One of the most impressive things about graphene is how fast electrons move through it. They travel through it more than 100 times faster than they do through the silicon used to make computer chips. In theory, this suggests that manufacturers could use graphene to make superfast transistors for faster, thinner, more powerful touch-screens, electronics, and solar cells.
But what makes graphene so amazing also hinders its use: Electrons flow through its honeycomb structure too easily. Unlike silicon, graphene lacks a bandgap. Bandgaps are the amount of energy an electron must gain to free itself from an atom and move to other atoms to conduct a current. Like a toll on a highway, electrons need to "pay" with energy to proceed. Electronic devices use bandgaps as gates to control where and when electrons flow. Lacking bandgaps, graphene's structure acts like an electron superhighway with no stop signs.
"Graphene's electrons are so wild and can't be tamed; it's hard to create a gap," said Andrei.
That lack of a bandgap makes graphene currently very difficult to use in modern electronics. Researchers supported by the Department of Energy's (DOE's) Office of Science are investigating ways to overcome this challenge and others to direct graphene's electron traffic.
Electrons Behaving Like Particles of Light
Materials that are just a few atoms thick act fundamentally different than larger amounts of the same material.
"The biggest challenge is having a reliable understanding of the properties of the materials," said Lilia Woods, a physics professor at the University of South Florida.
Even for a material that flat, graphene has some odd characteristics. In most materials, electrons move at different speeds. But in graphene, they all move at the same speed. In fact, electrons in graphene act as if they have no mass – like particles of light. That's one reason why the electrons move so fast and are so difficult to control.
Directing the Electron Traffic
Studying graphene's behavior is one thing. Figuring out how to manipulate it is another. Scientists have been pursuing several different ways to control the electrons in graphene: developing nanoribbons, stretching it, pairing it with boron nitride (another atom-tall material), and applying electrical charges to empty spaces in it. Scientists pursue multiple approaches because they don't know which one will work the best. In the meantime, each approach provides its own unique insight into graphene's basic properties.
Graphene Nanoribbons
Producing graphene nanoribbons is one way to make a material that's already unimaginably thin, even skinnier. These ribbons maintain many of graphene's positive characteristics while potentially giving scientists better control over how the electrons behave, including creating bandgaps.
"You can view these little ribbons as electronic circuit elements," said Michael Crommie, a physicist at DOE's Lawrence Berkeley National Laboratory (Berkeley Lab).
The investigation of nanoribbons began before scientists even got into the lab. Based on calculations, physicists theorized more than a decade ago that nanoribbons could offer new ways to manipulate graphene's electronic properties. Experimentalists have confirmed this idea by developing nanoribbons with consistent, clean edges.
For example, researchers at the University of Wisconsin and elsewhere developed graphene nanoribbons that displayed a bandgap. They showed that when the width of a nanoribbon is smaller than three nanometers, about the thickness of a strand of DNA, it develops a significant bandgap. It also becomes a semiconductor. Unlike graphene's electron superhighway, semiconductors can switch back and forth between conducting electricity or not. The narrower the ribbon, the bigger the gap or the "energy toll" electrons need.
But one challenge is how to make a single nanoribbon that has multiple widths and therefore regions with different bandgaps. Nanoribbons of a single width won't give scientists the level of control needed to design complex circuitry. To solve this problem, Berkeley Lab scientists fused together segments of ribbon with different widths. This "bandgap engineering" is essential to manufacturing semiconductor devices and a big step towards using graphene in circuits.

These nanoribbons can't be used by themselves, so scientists are currently investigating how nanoribbons interact with different surfaces. University of South Florida researchers studied graphene nanoribbons on silicon carbide (SiC) substrates. They found that how certain edges of nanoribbons attach to the SiC substrate influence the bandgap. Nanoribbons with different widths and edges anchored on different substrates can allow scientists more control over electron properties than nanoribbons that aren't anchored at all.
Stretching Graphene
Stretching graphene offers an alternative path for controlling its properties. When scientists stretch graphene in a specific way, it forms tiny bubbles in which electrons act as if they're actually in a very powerful magnetic field. These bubbles provide scientists with new opportunities for manipulating electron traffic in graphene.
This discovery was also a complete accident. A team at Berkeley Labhappened to be growing a layer of graphene on the surface of a platinum crystal in a vacuum chamber. As researchers tested the graphene, they noticed that its electrons were acting strangely. Rather than moving as they normally do in a smooth continuum, the electrons in the graphene nanobubbles bunched up at very specific energies. When researchers compared their results to what theory suggested, they found that the electrons were behaving as if they were in an ultra-strong magnetic field. However, there was no actual magnetic field present.
With graphene, "often we're chasing after one thing and we find something completely unexpected," Crommie said.
Pairing with Boron Nitride
When scientists first explored graphene's properties, they placed it on top of silicon dioxide. Because silicon dioxide is a common insulator for electronics applications, it seemed like an ideal match. However, the graphene wasn't reaching its full potential.
James Hone, a Columbia University mechanical engineering professor, recalled thinking, "Is there a layered material like graphene that would be a natural fit?"
Hone's team eventually discovered that graphene works much better when you put it on boron nitride instead. Like graphene, boron nitride can be made only a few atoms thick and has the same honeycomb structure. However, it's an insulator that impedes electrons from moving through it.
They found that putting boron nitride and graphene together can produce a new material whose properties are very flexible. This combination is so promising that Alex Zettl from Berkeley Lab joked that his lab is now "Boron Nitride R Us." He commented, "Having the boron nitride influence the graphene is a very powerful tool."
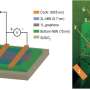
Ordinary light may offer a way to influence electrons in this new composite material. Berkeley Lab scientists have found that they can use light from a simple lamp to create an essential semiconductor device called a "p-n junction." P-n junctions have one side that's positive and lacks electrons and another side that's negative with extra electrons. By carefully designing these junctions, engineers can control how and when electrons move between the two sides of a material. They're like the gates that lift up and down at a toll booth.
Scientists realized that if they could put fixed, static charges in the boron nitride in a specific way, they could generate a p-n junction in the nearby graphene. To create the p-n junction, the scientists first prepared the graphene highway to have an excess of electrons, or be an n-type region. Then, by shining a light on the underlying boron nitride, they created a pothole, or p-type region, in the graphene. So with a light pulse and the boron nitride as a mediator, they could "write" p-n junctions – toll gates – into the graphene as needed.
Even after scientists turned off the light, the activation of the boron nitride and its influence on the electron traffic in the nearby graphene, stayed in place for days. The scientists also discovered that they could erase and re-create these junctions, which could be important for designing electronic devices.
Now researchers are using scanning tunneling microscopes, which use nanometer-sized tips to conduct electricity, to do the same thing with more precision.
Charging Up Empty Spaces in Graphene
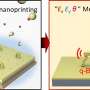
Because of its unique structure, graphene remains stable even when scientists punch holes in it. Andrei's team from Rutgers University took advantage of this fact to create an "artificial atom" that influences nearby electrons in the undamaged part of graphene. First, researchers shot helium at graphene on a substrate, knocking out a single carbon atom. They then used a scanning tunneling microscope to apply a positive charge to the substrate under the empty space where the missing atom used to sit. Like a real atom, that positive charge influenced the orbits of electrons in the surrounding graphene. Creating these artificial atoms could be another way that future devices could control electron flow in graphene.
The Future of Graphene
Perhaps the most surprising of these twists and turns is that the future may not lie in graphene at all. As scientists investigated graphene's unique electronic properties, they discovered new extremely thin materials made from elements other than carbon. If a material is only a few atoms thick and has a honeycomb structure, it can demonstrate many of graphene's electronic properties. In fact, scientists have found materials made of silicon, germanium, and tin that act strikingly similar to graphene. Using these materials by themselves or in combination with graphene may offer better characteristics than graphene alone.
In the meantime, scientists will continue to investigate the strange features of this frequently surprising material. As Philip Kim, a Harvard University physics professor said, "[Graphene] always provides you with some new, exciting science that we have not expected."
Provided by US Department of Energy