Engineers pioneer platinum shell formation process, achieve first-ever observation
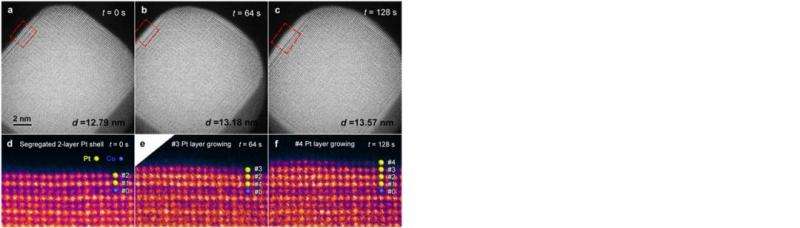
UCI researchers have devised a new method of dynamically forming a platinum shell on a metallic alloy nanoparticle core, a development that may lead to better materials for oxygen reduction reactions in fuel cells that power some cars and electronic devices. In a first, engineers were able to observe the process directly, in real time, in UCI's state-of-the-art transmission electron microscopy facility, part of the recently established Irvine Materials Research Institute.
Reported in Nature Communications, the method centers on an oxygen annealing treatment to enrich the concentration of the metal on the platinum-cobalt nanomaterial core, a more effective procedure than traditional acid leaching or reductive annealing, according to the research team.
"A deeper understanding of platinum shell formation is critical for synthesis and structural control of platinum-metallic nanoparticles," said team leader Xiaoqing Pan, Henry Samueli Endowed Chair in Engineering and professor of chemical engineering & materials science at UCI. "We were able to achieve atomic-scale observation of the reaction process at atmospheric pressures in our TEM, something that has never been successfully accomplished until now."
Pan, who's also the inaugural director of the Irvine Materials Research Institute and a professor of physics & astronomy, said his group's work "may open a new avenue to study gas-solid interactions at atomic scale under the atmospheric pressure at which many real gas-phase reactions take place."
The project was supported by IMRI and the National Science Foundation.
More information: Sheng Dai et al. In situ atomic-scale observation of oxygen-driven core-shell formation in Pt3Co nanoparticles, Nature Communications (2017). DOI: 10.1038/s41467-017-00161-y
Journal information: Nature Communications
Provided by University of California, Irvine