Researchers create one-nanometer trimetallic alloy particles
The principal components of petroleum and natural gas are hydrocarbons and their mixtures, indispensable as resources supporting modern infrastructure as raw materials for the petrochemical industry. A technique conventionally used to create beneficial chemical products from hydrocarbons is using a large amount of metallic peroxides in hazardous organic solvents to oxidize hydrocarbon compounds.
To use resources effectively and to reduce environmental impact, clean catalytic oxidization without solvents using the oxygen in the air has been a popular research subject in recent years. Research of noble metal nanoparticles supported on porous carbon materials or metallic oxides are especially prevalent, and they are viewed as promising catalysts. Vital elements determining the reactivity of such heterogeneous catalysts are the shape, size, and metallic composition of the metallic nanoparticles. Particles less than 2 nm are important for new high-performance catalysts, since reducing the diameter of the catalyst particle not only increases the surface area ratio but greatly changes the state of the electrons on the surface of the metals, in turn greatly changing the reactivity. However, finding a method of synthesizing these small metallic nanoparticles while controlling both the diameter and composition is challenging.
Overview
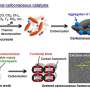
The research group led by Kimihisa Yamamoto of Tokyo institute of Technology developed a method of synthesizing microscopic alloy nanoparticles using branched molecular dendrimers developed in the Yamamoto Atom Hybrid Project on the ERATO program. Molecules called dendrimers have a regular branching structure with only one definite molecular weight, although they are classified as macromolecules. The research group implemented many coordination sites for forming metal ions and complexes. By using a dendrimer with such coordination sites as a template for the nanoparticle, the group was able to synthesize a nanoparticle with a controlled number of atoms.
Further, they evaluated the activity of this alloy nanoparticle as an oxidization catalyst for hydrocarbons under ordinary pressures when using oxygen in the air as the oxidizing agent. They found that its activity was 24 times greater than that of commercially available catalysts for oxidization of organic compounds. They also found that by adding a catalytic amount of organic hydroperoxide, this catalyst promotes the oxidization of hydrocarbon into aldehydes and ketones under ordinary temperatures and pressures. Further, by comparing the changes in activity due to alloy catalysts of different metallic compositions and examining the composition and other characteristics of the intermediates, ketones and organic hydroperoxides, the group was able to observe the process of reaction promotion due to the alloying of the catalyst.
Future Development
The knowledge gained from this research could contribute to design guidelines for new high-performance catalysts. The method for synthesizing alloy nanoparticles developed in this research can be used generally and applied to other metals. For this reason, this could be the technology uncovering the reactivity of other microscopic alloy nanoparticles. Further study is required on the increase of catalytic activity at the interface of copper and other noble metals in the oxidizing transformations of other organic compounds, not only the oxidization of hydrocarbons. Application is anticipated for next-generation high-performance materials in the fields as diverse as optics, electronics, and energy.
More information: Masaki Takahashi et al, Finely controlled multimetallic nanocluster catalysts for solvent-free aerobic oxidation of hydrocarbons, Science Advances (2017). DOI: 10.1126/sciadv.1700101
Journal information: Science Advances
Provided by Tokyo Institute of Technology