Innovative carbon nanotube photocatalytic materials for efficient solar energy conversion and hydrogen production
The unique properties of semiconducting single-walled carbon nanotubes (s-SWCNTs) offer significant advantages over organic molecules, semiconducting polymers, and solid-state semiconductors for wide ranging applications. In particular, s-SWCNT are potentially highly effective active absorption layers in thin-film solar cells because the optical absorption bands—that depend on the chiral indices, (n,m)—of s-SWCNTs exhibit excellent overlap with the useful solar spectrum range of the sun's radiation.
However, in other applications of carbon nanotubes, there have not been any reports on the use of s-SWCNTs as components of photocatalysts for splitting water into hydrogen (photocatalytic H2) although photocatalytic-based water splitting is expected to be a key technology for solar energy conversion and sustainable production of hydrogen.
Now, Yutaka Takaguchi and colleagues at Okayama University, Yamaguchi University, and Tokyo University of Science report on the observation of photocatalytic H2 evolution from water triggered by photoexcitation of s-SWCNTs.
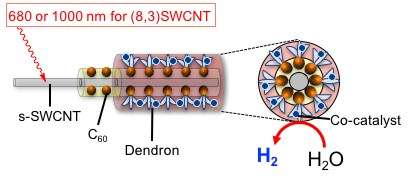
The researchers fabricated a structure consisting of a s-SWCNT/C60 coaxial heterojunction by a self-organization technique using fullerodendron to make s-SWCNT act as a photocatalyst. This heterojunction was used to induce the highly efficient H2 evolution reaction from water, where the (8, 3) SWCNT/fullerodendron coaxial photocatalyst shows H2-evolving activity (QY = 0.015) upon 680-nm illumination, which is E22 absorption of (8, 3) SWCNT.
Due to strong absorption coefficients and ease of modifying s-SWCNTs, the CNT-photocatalyst could be a powerful candidate as a material for solar energy conversion and H2 production without CO2 emission.
More information: Noritake Murakami et al. SWCNT Photocatalyst for Hydrogen Production from Water upon Photoexcitation of (8, 3) SWCNT at 680-nm Light, Scientific Reports (2017). DOI: 10.1038/srep43445
Journal information: Scientific Reports
Provided by Okayama University



















