July 18, 2017 report
A way to speed up reaction discoveries using automation
(Phys.org)—A pair of researchers with the University of California and Lawrence Berkeley National Laboratory has found a way to automate reaction discoveries, thereby speeding up the process. In their paper published in the journal Science, Konstantin Troshin and John Hartwig describe their process, how well it works, and a discovery they have already made using it.
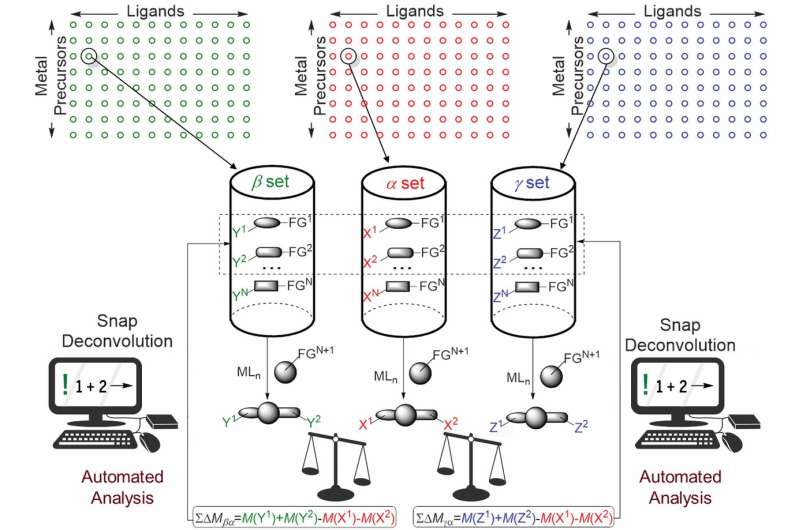
Discovering new chemical reactions with useful applications is generally a laborious process. It typically involves combining large groups of reagents two at a time with a catalyst then studying the molecular makeup to see what might be useful—the more experiments conducted, the more likely chemists will find something new and useful. This situation is unfortunate, because the discovery of new reactions leads to the development of new products. This is why some chemists seek ways to automate at least some of the process. In this new effort, the researchers describe a technique that involves the use of a gas chromatography/mass spectrometry GC/MS device, a sealed 96-well plate, and a slew of spreadsheet macros.
In general, the new technique involves loading up the wells with reactants, adding different amounts of ligand and metal combinations, and heating them to 100°C for 18 hours. That is followed up by using GC/MS to observe changes in each of the wells and analyzing the results using spreadsheet macros. More specifically, the technique involves starting with just three compounds divided into pools that belong to the same type of molecular function group. Before the reactions are induced, inert substitutes are added, each varying in mass. When the reactions occur, the unique mass of each allows for easy detection by GC/MS and subsequent analysis using the macros.
The researchers report that their technique speeds up the process used for discovering useful new reactions by a factor of three by automating some of the time-consuming steps. They claim it works as advertised, noting that they used it to discover a three-component, nickel-catalyzed diarylation of alkynes.
More information: Snap deconvolution: An informatics approach to high-throughput discovery of catalytic reactions , Science 14 Jul 2017: Vol. 357, Issue 6347, pp. 175-181, DOI: 10.1126/science.aan1568 , science.sciencemag.org/content/357/6347/175
Abstract
We present an approach to multidimensional high-throughput discovery of catalytic coupling reactions that integrates molecular design with automated analysis and interpretation of mass spectral data. We simultaneously assessed the reactivity of three pools of compounds that shared the same functional groups (halides, boronic acids, alkenes, and alkynes, among other groups) but carried inactive substituents having specifically designed differences in masses. The substituents were chosen such that the products from any class of reaction in multiple reaction sets would have unique differences in masses, thus allowing simultaneous identification of the products of all transformations in a set of reactants. In this way, we easily distinguished the products of new reactions from noise and known couplings. Using this method, we discovered an alkyne hydroallylation and a nickel-catalyzed variant of alkyne diarylation.
Journal information: Science
© 2017 Phys.org