Large single-crystal graphene is possible
Thanks to its conductivity, strength and flexibility, graphene is considered as one of the most likely substitutes for silicon and other materials. However, it has not yet resulted in industrial applications. High-quality single-crystal graphene can only be produced in quantities a couple of millimeters to mere centimeters in size. Recently, a team led by Prof. Feng Ding and Prof. Rodney Ruoff with the Institute for Basic Science (IBS), Prof. Kaihui Liu at Peking University, and their collaborators reported the synthesis of a large sheet of monolayer single-crystal graphene. This result allows a leap forward in graphene production to an optimized method of fabricating an almost-perfect (> 99.9 % aligned) 5 × 50 cm2 single-crystal graphene in just 20 minutes. Moreover, the low production costs, comparable to commercially available lower-quality polycrystalline graphene films, could expand its usability. Detailed in Science Bulletin, the method is expected to stimulate further fundamental work on graphene and related materials, including large-scale folding of graphene sheets, similar to paper, creating origami-like or kirigami-like shapes that could be applied to future flexible circuits.
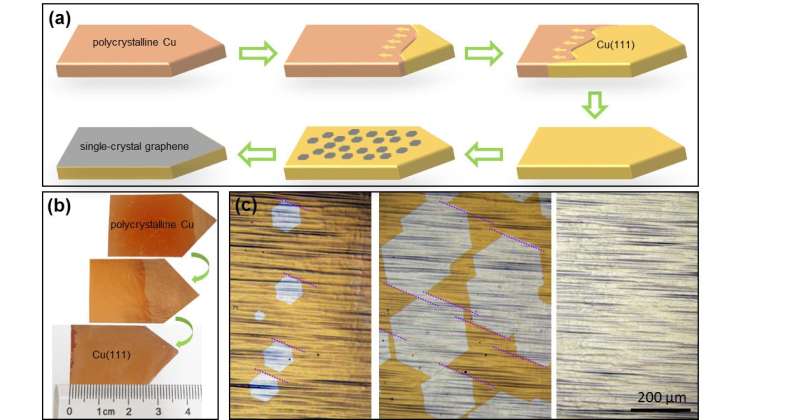
Single-crystal graphene is a honeycomb-shaped monolayer of carbon atoms, uniform throughout the material On the other hand, polycrystalline graphene is formed by randomly oriented graphene islands, which decrease its quality. Currently, scientists are able to grow meter-sized polycrystalline graphene and smaller single-crystal graphene, ranging from 0.01 mm2 to a few cm2. The synthesis of large single-crystal graphene at a low cost is a critical goal of graphene synthesis. In this study, graphene is grown on the surface of a 5 × 50 cm2 copper foil, which was transformed into a single-crystal copper foil by heating to ~ 1,030°. The temperature slope from hot to cold moved the so-called grain boundary onwards, creating a perfect single crystal. In the heating and cooling treatment, copper atoms migrate inside the material, arranging into an ordered structure with fewer defects. "The secret to obtaining single-crystal graphene at large sizes is to have a perfect single-crystal copper as a base to start with. Large single-crystal copper foil is not commercially available, so labs must build it with their own means," explains Feng Ding, group leader at the Center for Multidimensional Carbon Materials.
Then, via another technique called chemical vapor deposition, millions of parallel graphene islands are formed on the copper foil surface. As more carbon atoms are deposited on the foil, the islands grow until they coalesce and form a near-perfect single-crystal graphene layer covering the entire available surface.

In order to optimize the technique, the team had to consider four technical challenges: (i) preparation of single-crystal copper foil in a very large area, (ii) obtaining a high degree of alignment of the graphene islands following their nucleation and growth, (iii) seamlessly stitching the graphene islands into a single crystal through further growth and (iv) the fast growth of single-crystal graphene. Although previous reports have addressed some of the above challenges, this study overcame all of them and made the synthesis of meter-sized single-crystal graphene possible. The degree of the misaligned graphene islands is less than 0.1 percent, amounting to negligible defects and grain boundaries in the products.
The current result was limited only by the size of the copper foil and, in principle, both the size of the copper foil and graphene film could be unlimited. In addition, considering the very short time for graphene synthesis (20 minutes) and the relatively low-cost experimental setup, the price of a single-crystal (or nearly single-crystal) graphene could be close to that of current polycrystalline graphene films.
"The dream of many scientists is to replace silicon," says Ding. "Now, we are exploring which is the best material to grow graphene on top, and how to use copper as a substrate for other interesting 2-D materials."
More information: Xiaozhi Xu et al, Ultrafast epitaxial growth of metre-sized single-crystal graphene on industrial Cu foil, Science Bulletin (2017). DOI: 10.1016/j.scib.2017.07.005
Provided by Institute for Basic Science