Longer-lasting pain relief with MOFs
To treat headaches, back pain or fever, most of us have reached for ibuprofen at one point or another. But we often have to take doses every four to six hours if the pain warrants it. Now scientists are working on a way to package the commonly used drug so it can last longer. Their approach, reported in ACS' journal Molecular Pharmaceutics, could also be used to deliver other drugs orally that currently can only be taken intravenously.
Recently, scientists have been studying compounds called metal-organic frameworks (MOFs), which are made of metal ions linked to organic ligands, for drug delivery. Active ingredients can be packed inside MOFs, which are porous, and some of them have additional traits such as water solubility that make them good candidates for drug couriers. But few studies have so far investigated whether such MOFs could be used in oral formulations. J. Fraser Stoddart and colleagues wanted to test promising MOFs using ibuprofen as a model drug.
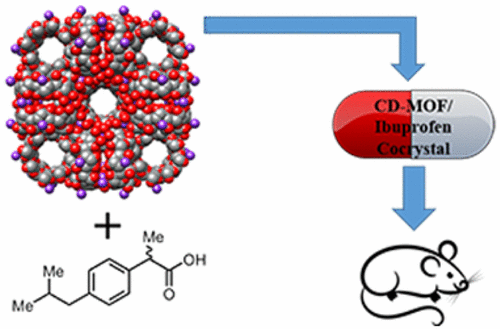
The researchers loaded therapeutically relevant concentrations of ibuprofen into easily prepared, biocompatible MOFs with cyclodextrin and alkali metal cations. Testing in mice showed that the compounds reached the blood stream quickly in about 10 to 20 minutes and lasted twice as long as ibuprofen salts, which are the active ingredient in commercial liquid gel formulations. The researchers say the promising findings suggest that these compounds could take the next step toward commercial development for delivering ibuprofen and potentially other drugs.
More information: Karel J. Hartlieb et al. Encapsulation of Ibuprofen in CD-MOF and Related Bioavailability Studies, Molecular Pharmaceutics (2017). DOI: 10.1021/acs.molpharmaceut.7b00168
Abstract
Although ibuprofen is one of the most widely used nonsteroidal anti-inflammatory drugs (NSAIDs), it exhibits poor solubility in aqueous and physiological environments as a free acid. In order to improve its oral bioavailability and rate of uptake, extensive research into the development of new formulations of ibuprofen has been undertaken, including the use of excipients as well as ibuprofen salts, such as ibuprofen lysinate and ibuprofen, sodium salt. The ultimate goals of these studies are to reduce the time required for maximum uptake of ibuprofen, as this period of time is directly proportional to the rate of onset of analgesic/anti-inflammatory effects, and to increase the half-life of the drug within the body; that is, the duration of action of the effects of the drug. Herein, we present a pharmaceutical cocrystal of ibuprofen and the biocompatible metal–organic framework called CD-MOF. This metal–organic framework (MOF) is based upon γ-cyclodextrin (γ-CD) tori that are coordinated to alkali metal cations (e.g., K+ ions) on both their primary and secondary faces in an alternating manner to form a porous framework built up from (γ-CD)6 cubes. We show that ibuprofen can be incorporated within CD-MOF-1 either by (i) a crystallization process using the potassium salt of ibuprofen as the alkali cation source for production of the MOF or by (ii) absorption and deprotonation of the free-acid, leading to an uptake of 23–26 wt % of ibuprofen within the CD-MOF. In vitro viability studies revealed that the CD-MOF is inherently not affecting the viability of the cells with no IC50 value determined up to a concentration of 100 μM. Bioavailability investigations were conducted on mice, and the ibuprofen/CD-MOF pharmaceutical cocrystal was compared to control samples of the potassium salt of ibuprofen in the presence and absence of γ-CD. From these animal studies, we observed that the ibuprofen/CD-MOF-1 cocrystal exhibits the same rapid uptake of ibuprofen as the ibuprofen potassium salt control sample with a peak plasma concentration observed within 20 min, and the cocrystal has the added benefit of a 100% longer half-life in blood plasma samples and is intrinsically less hygroscopic than the pure salt form.
Journal information: Molecular Pharmaceutics
Provided by American Chemical Society