Cleaner, more durable surface coatings through collaborative study of electrostatics
Work surfaces at home that we clean regularly still accumulate mystery particulate matter over time: sinks, tiles, and windows—even non-stick Teflon pans. The same is true of the surfaces of materials used in industry and in environmental remediation. Efforts to create better materials with predictable surface properties have led scientists to study the electrostatic forces that attract particulates to surface coatings. Recently, Pacific Northwest National Laboratory's (PNNL's) Hongfei Wang collaborated with a team of researchers from Northwestern University (NU) to dramatically advance the ability to study the electrostatic charges of surfaces. The team's innovative laser-based technique now allows researchers to predict how a surface's electrostatic charge might attract particulates with opposite charges, decreasing the buildup of what NU's Franz Geiger calls "crud."
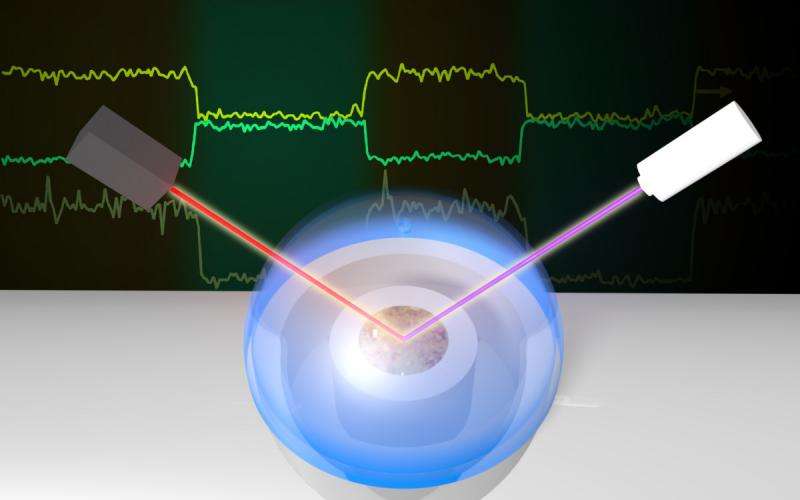
"Weak bonds, and many of them, are what cause particulates to stick," said Geiger. "Like Velcro, tons of tiny, weak hooks establish bonds that bind crud to surfaces. But because there are so many electrostatic connections, they become difficult to pry apart." Studying these electrostatic forces required researchers to devise an improved optical approach and to innovate to overcome hurdles, such as the presence of water and mineral-structure effects that impeded precise measurements. "It's hard to study surfaces under water because most available surface science techniques require vacuum conditions, under which water evaporates or freezes," explains Geiger. "Our techniques overcome these problems using laser-based technologies that probe interfaces of interest by focusing an intense light source on the surface and measuring changes in the light collected after interaction. Because the signals are only generated where symmetry is broken—just like at an interface between water and a solid—we are able to gain the information needed," said Geiger. Through continual innovation, the team figured out how to isolate the surface response from the bulk response at the alpha-quartz/water interface. This feat is similar to isolating just one cheering voice in a stadium packed full of screaming fans.
Generally hidden from our perception without advanced scientific instrumentation, electrostatic forces attract fine bits of particulates that cement onto surfaces, creating costly problems. It's far easier to find ways to remove "crud" by scrubbing kitchen tiles with abrasives than it is to remodel a kitchen, but it's still costly in terms of time and elbow grease spent. The same holds true in industry and in environmental remediation, where costs from fixing or replacing fouled surfaces can be measured in years and many millions of dollars. It is therefore essential to create materials with surface coatings equipped to stand the tests of time and chemistry.
The researchers' new technique outcompetes other approaches, which cost significantly more and are slower and less adaptable to the shapes and designs of consumer and industrial materials. Eventual applications could include improved surface coatings for products from sunglasses to mobile phones to ceramics and laminates. Among many others, the new technique also presents broad potential applications in environmental remediation projects, including U.S. DOE legacy sites at Hanford, Savannah River, and Oak Ridge.
The development of the researchers' successful technique was the result of an intense trial-and-error process. They started with the experimental set up traditionally used for second harmonic generation studies in Geiger's group, which included a femtosecond laser oscillator from Newport Spectra Physics, optical lenses and mirrors, and a flow cell produced in-house for housing the alpha-quartz/water interface. A detector connected to a signal amplifier and computer recorded the signal. The researchers progressed this setup through a multi-year series of iterations before landing on their new design, which upends the traditional method by probing the solid surface from the aqueous side. Challenges included overcoming experimental design problems, such as creating a reliable method to monitor the miniscule energy fluctuations in their light source that would show up as interference in the measured signal. The end result of their responses to these challenges was a massive suppression of the bulk response from the quartz, enabling the faint surface signal to be detected with exquisite sensitivity (see sidebar). The result is good news for geochemists because alpha-quartz is the most abundant silicate mineral in the environment.
The researchers' work connects tightly to the ongoing field-work project ERKCC72 at Oak Ridge National Laboratory, "Atomic- to Pore-Scale Geochemical Processes," led by Andrew G. Stack. That project aims to improve our atomistic understanding of mineral/water interactions in nature, which now opens the possibility to understand the alpha-quartz/water interface by pairing the experiments to atomistic calculations. "We are very excited about this prospect," said Geiger. "At the end of the day, environmental cleanup is really expensive, involving multi-million-dollar projects that require the clean-up chemistry to work for a long time."
More information: Paul E. Ohno et al. Phase-referenced nonlinear spectroscopy of the α-quartz/water interface, Nature Communications (2016). DOI: 10.1038/ncomms13587
Journal information: Nature Communications
Provided by Pacific Northwest National Laboratory