New 'biomimetic' glue shows high-strength bonding under water
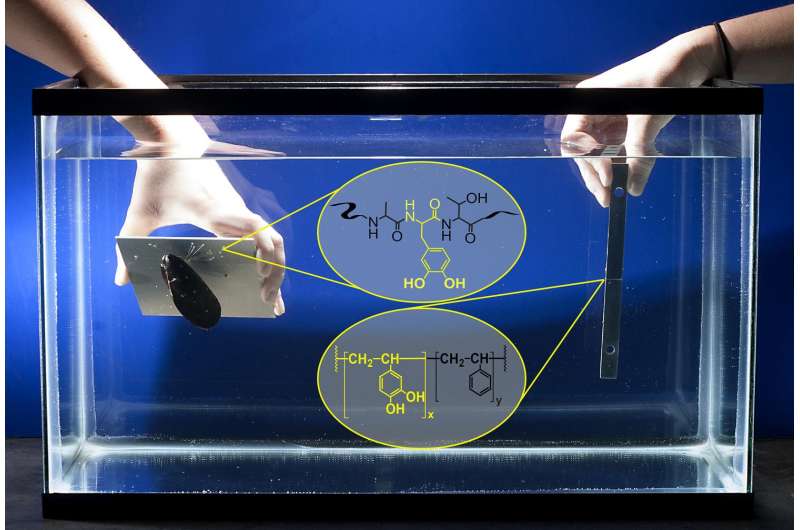
An adhesive that works under water and is modeled after those created by shellfish to stick to surfaces is stronger than many commercial glues created for the purpose.
"Our current adhesives are terrible at wet bonding, yet marine biology solved this problem eons ago," said Jonathan Wilker, a professor of chemistry and materials engineering at Purdue University. "Mussels, barnacles, and oysters attach to rocks with apparent ease. In order to develop new materials able to bind within harsh environments, we made a biomimetic polymer that is modeled after the adhesive proteins of mussels."
New findings showed that the bio-based glue performed better than 10 commercial adhesives when used to bond polished aluminum. When compared with the five strongest commercial glues included in the study, the new adhesive performed better when bonding wood, Teflon and polished aluminum. It was the only adhesive of those tested that worked with wood and far out-performed the other adhesives when used to join Teflon.
Findings are detailed in a research paper published online in February and in the March 1 print issue of the journal ACS Applied Materials and Interfaces.
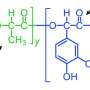
Mussels extend hair-like fibers that attach to surfaces using plaques of adhesive. Proteins in the glue contain the amino acid DOPA, which harbors the chemistry needed to provide strength and adhesion. Purdue researchers have now inserted this chemistry of mussel proteins into a biomimetic polymer called poly(catechol-styrene), creating an adhesive by harnessing the chemistry of compounds called catechols, which are contained in DOPA.
"We are focusing on catechols given that the animals use this type of chemistry so successfully," Wilker said. "Poly(catechol-styrene) is looking to be, possibly, one of the strongest underwater adhesives found to date."
While most adhesives interact with water instead of sticking to surfaces, the catechol groups may have a special talent for "drilling down" through surface waters in order to bind onto surfaces, he said.
The series of underwater bond tests were performed in tanks of artificial seawater.
"These findings are helping to reveal which aspects of mussel adhesion are most important when managing attachment within their wet and salty environment," Wilker said. "All that is needed for high strength bonding underwater appears to be a catechol-containing polymer."
Surprisingly, the new adhesive also proved to be about 17 times stronger than the natural adhesive produced by mussels.
"In biomimetics, where you try to make synthetic versions of natural materials and compounds, you almost never can achieve performance as good as the natural system," Wilker said.
One explanation might be that the animals have evolved to produce adhesives that are only as strong as they need to be for their specific biological requirements. The natural glues might be designed to give way when the animals are hunted by predators, breaking off when pulled from a surface instead of causing injury to internal tissues.
The research paper was authored by former doctoral students Michael A. North and Chelsey A. Del Grosso, who have graduated, and Wilker.
Future research will include work to test the adhesive under real-world conditions.
"We have shown that this adhesive system works quite well within controlled laboratory conditions. In the future we want to move on to more practical applications in the real world," Wilker said.
More information: Michael A. North et al. High Strength Underwater Bonding with Polymer Mimics of Mussel Adhesive Proteins, ACS Applied Materials & Interfaces (2017). DOI: 10.1021/acsami.7b00270
Journal information: ACS Applied Materials and Interfaces
Provided by Purdue University